|
Professor J.-C.G. Bünzli is a physical-inorganic and analytical chemist by training and an active researcher in the field of co-ordination and supramolecular chemistry of the lanthanide ions. His research focuses mainly on the relationship between the luminescent properties of lanthanide ions and the structure of the edifices in which these ions are embedded, on the use of lanthanide ions (mainly europium and terbium) as luminescent probes for biomedical analyses, and on the design of self-assembled building blocks for the synthesis of materials with predetermined photophysical and magnetic properties. The design of receptors able to encapsulate lanthanide ions and of enhancing their physico-chemical properties follows four different approaches. |
 |
|
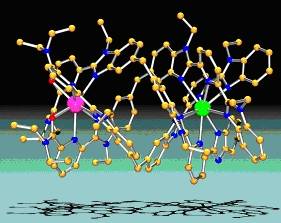
| 1) Macrocyclic receptors The lock-and-key principle is of little use with lanthanide ions which display very similar chemical properties. As a consequence, two classes of macrocycles, namely cyclens and calixarenes, are modified by grafting functionalized arms on their framework. These arms fold upon complexation and form an induced cavity (Inorg. Chem. 2002, 2459 ; J. Chem. Soc. Dalton Trans. 2001, 3205).2) Podands Here the principle is the same as above, but the macrocyclic core is replaced either by a single atom (nitrogen, boron) or by a small aromatic ring (benzene). This strategy allowed us to produce lanthanide podates which self-assemble to yield nanoparticles with interesting properties for medical imaging via magnetic (J. Am. Chem. Soc. 2000, 122, 10810).3) Synthons based on aromatic tridentate units One common coordination number for lanthanide ions is nine. Therefore, the use of three tridentate ligands leads to the formation (often by strict self-assembly) of triple helical complexes in which the coordination requirements of the metal ion are satisfied. These edifices display chiral properties that may be taken advantage of in the design of chiral luminescent probes. Three classes of ligands are being developed, based on bis(benzimidazole)pyridine, terpyridine and dipicolinic acid (J. Chem. Soc. Dalton Trans. 2000, 2809 et 2001, 2655 ; Inorg. Chem. 2002, 2642). 4) Self-assembly of dimetallic triple-.stranded helicates In addition the various energy migration processes intervening in the sensitization of a lanthanide ion luminescence via ligand states are being studied in great detail in our laboratory, both experimentally and from a theoretical point of view (J. Phys. Chem. A 2002, 106, 1670). Finally, grafting long hydrophobic arms onto coronands or bis(benzimidazole)pyridines allows us to engineer metallomesogenic structures, that is liquid crystals incorporating metal ions (in collaboration with Professor C. Piguet from the University of Geneva, cf. Chemistry of Materials, 2002, 14, 5705). |
||
 |
Time-resolved lanthanide luminescence for lab-on-a-chip detection of biomarkers on cancerous tissues. A combination of microfluidics, lanthanide luminescent bioprobes and hystoimmunochemical chemistry allows analyses to be carried out in 5-6 less time and with 5-time less reactants than through the conventional method.Analyst, 2009, 134, 1991 – 1993 |
 |
Lanthanide luminescence for functional materials and bio-sciences. Svetlana V. Eliseeva and Jean-Claude G. Bunzli.Chemical Society Reviews, Vol. 39, Nu. 1, Jan. 2010, p. 1-380 |