Structure and dynamics of biomolecules studied by nuclear magnetic resonance
 |
Two research groups in Paris and Lausanne are working in complementary fashion on fundamental aspects of nuclear magnetic resonance (NMR) spectroscopy and on a variety of applications to biological macromolecules such as proteins and nucleic acids (DNA or RNA). In NMR spectroscopy, it is possible to design an incredible variety of experimental methods that are applicable to liquids, to liquid crystals, and to amorphous or polycrystalline solids. | |
| Suitable methods can be designed to measure parameters that provide information about molecular structure (internuclear distances, dihedral angles, orientation of certain chemical bonds with respect to a common frame, etc.), about intramolecular dynamics (rearrangements, tautomerism, local mobility, internal entropy, local order and disorder), about rotational diffusion (molecular tumbling) and about translational diffusion. The range of applications of modern NMR is truly staggering, since it encompasses essentially all materials ranging from rocks to polymers, from crystals to glasses, from catalysts to enzymes, from cell membranes to biological transport phenomena, from pharmaceuticals to the tracking of metabolites (“metabonomics”). | ||
Methodology
 |
Modern applications of NMR to biomolecules, either in solution or in the solid phase, require methods that aim at manipulating the magnetization of nuclei such as protons, deuterons, carbon-13, nitrogen-15, or phosphorus-31. Such experiments involve radio-frequency pulses with different frequencies, phases and time-dependent amplitudes, combined with brief intervals where magnetic field gradients are applied to the sample. The use of magnetic fields that depend on the spatial coordinates within the sample allows one to distinguish signals that depend on the spatial localisation of the molecules, in the manner of magnetic resonance imaging (MRI). Subtle features arise from the interplay between components of the magnetization from different parts of the sample (“dipolar fields”) and from the coupling of these components to the receiving antenna (“radiation damping”). The design of novel experiments presupposes a good grasp of the underlying theory, which, although rooted in quantum mechanics, is readily accessible to a wide audience of physicists, chemists and biologists, because one can benefit from elegant concepts such as “coherence transfer”. Simple rules allow one to design methods for the excitation and observation of coherences. Reliable theoretical tools allow one to predict the life-times of various coherences. In solution-state NMR, the measurement of these life-times (“relaxation times”) provides valuable information on both the average molecular structure and internal dynamics: In particular, so-called “cross-correlation effects” can provide insight into the relative orientations of chemical bonds. Further information about such parameters can be obtained from NMR in the solid state, where it is possible to remove certain undesirable effects by rapid rotation of the sample (“magic angle spinning” or MAS) and then deliberately reintroduce the effect of selected interactions that provide information on distances between nuclei. Such measurements provide insight that is by essence complementary to structures obtained from X-ray or neutron diffraction. The measurement of translational diffusion provides a measure of the “bulkiness” of a macromolecule or of a complex involving several molecules. Many other types of measurements can be conceived that provide insight into the life of molecules on an atomic level. |
Intermolecular affinity
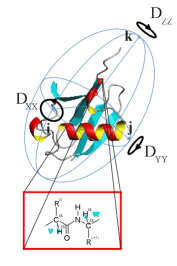 |
One of the long-term goals of our current research is to obtain a better understanding of the affinity between two molecules, be it between two biological macromolecules such as a protein and a nucleic acid, or between a protein on the one hand, and a small ligand such as an inhibitor, a cofactor, or a substrate on the other hand. The affinity of two partners that make a complex can be expressed in terms of an association constant. Intermolecular affinity is usually rationalized in terms of enthalpic factors (hydrogen bonds, electrostatic effects, etc.) and entropic factors due to the relative immobilisation of the two partners, to the ejection of solvent molecules from the interface, etc. However, it is possible that intermolecular affinity is strongly affected by the internal mobility of macromolecules. Indeed, the affinity could be enhanced if the encounter between a macromolecule and its target were accompanied by a sort of ‘internal melting’ of the macromolecule, i.e., by an increase of entropy due to internal degrees of freedom. A gain in intramolecular mobility could turn out to be one of the driving forces of intermolecular affinity. Such a hypothesis can only be verified if one develops tools that allow one to measure internal entropy, or at least to measure the extent of intramolecular mobility. We have therefore developed several novel methods that allow one to characterise the mobility of side-chains and of the backbone of proteins, and of hydrogen bonds in proteins and in nucleic acids. |
An unusual type of research
|
In practice, the activity in our research laboratory is quite distinct from typical procedures used in chemical or biochemical laboratories. We do not carry out any chemical synthesis, nor do we clone, grow bacteria, express proteins or purify samples all of these essential things are done in other laboratories who kindly provide us with samples. Many ideas for new experiments are often based on some loose intuition, sometimes inspired by work in other laboratories. When these poorly-designed experiments are tried out in quick-and-dirty attempts, they often turn out to provide unsatisfactory results. The failures and shortcomings of such initial experiments provide inspiration for a study of possible imperfections and for a deeper theoretical analysis. This often involves extensive simulations of the behaviour of spin systems. The experiments are then gradually refined. In some cases, we have been able to reduce systematic errors by as much as two orders of magnitude! |