
Activities involving biological material are categorized into four classes, often referred to as BioSafety Levels (BSL), based on factors such as the hazard level of the organism, the scale of the activity, and the specific procedures involved.
At EPFL, there are BSL 1, BSL 2, and BSL 3 laboratories; however, BSL 4 laboratories are not present.
Experiments involving biological material at EPFL are regulated by the Technical Supplementary Directive to LEX 1.5.1: Complementary Technical Directive to LEX 1.5.1: Safety Concept as Defined by the OUC and OPTM for Laboratories Working with Biological Material. This safety concept is available on the dedicated EPFL OHS Directives web page.
As defined by the ContainO ordinance, activities involving:
• genetically modified organisms
• pathogenic organisms
• exotic (alien) organisms
must:
• be conducted under mandatory containment
• be supervised by a designated Biosafety Officer (BSO)
• be notified to or authorized by the federal authorities
The EPFL biosafety team prepares these documents for the search groups; however, the Principal Investigators (PIs) and the BSO are responsible for providing the biosafety team with the necessary information to draft these notifications and authorization documents. They must also inform the biosafety team of any new organism (cell line, bacterium, virus, etc.) and any new activity planned in their research unit.
For any support, or complementary information, contact us.
For information on biological hazard training courses, please visit our dedicated webpage.
In case of accident
Call the emergency number 115 or +41 21 693 30 00
Class 1 activities involve well-characterized agents not known to consistently cause diseases in immunocompetent individuals. These activities present minimal potential hazard to laboratory personnel and the environment (no or low individual and community risk).
In this way, biosafety level 1 (BSL 1) laboratories are not necessarily separated from the other labs in the building. Work is typically conducted on open bench tops using standard microbiological practices. Special containment equipment is not required, but may be used as determined by appropriate risk assessment.
Guidelines for Biosafety Level 1 laboratory
You have to declare class 1 activities to authorities when genetically modified material is involved (modified cells, bacteria, animals, plants, etc.).
To establish if a material is genetically modified you can check the Annex 1 of the “Ordinance on Handling Organisms in Contained Systems”. The biosafety team will help you with the declaration.
Training:
The online course “Working in a biosafety level 1 laboratory” provides an overview of the main hazards that can be encountered in a biosafety level 1 laboratory, and covers topics such as good microbiological practice, accident management and chemical and biological waste management.
Working in a biosafety level 1 laboratory
Question about BSL 1 waste? We have a dedicated webpage for Biological waste management.
BSL 2 is suitable for work involving agents that can cause human or animal disease but is unlikely to be a serious hazard to laboratory workers, the community, livestock or the environment. Laboratory exposures may cause serious infection, but effective treatment and preventive measures are available and the risk of spread of infection is limited.
For BSL 2 environments:
- a specific training is provided for laboratory personnel
- the access is restricted
- a biohazard sign is placed on the lab door
- you have to minimize aerosol production and know how to correctly use a biosafety cabinet
All procedures in which aerosols or splashes may be created are conducted in a biosafety cabinet.
In a BSL2 lab, waste must be inactivated before disposal.
Guidelines for Biosafety Level 2 laboratory
Guidelines for Prion-like proteins
You have to declare class 2 activities to authorities. The biosafety team will help you with the declaration.
Any question about BSL 2 waste ? We have a dedicated webpage for Biological waste management.
Please register for the FOBS 3 training and ask for the access rights via AxS.
The workflow for the validation of the BSL 2 laboratory access is the following:
- The person requests the access to the BSL 2 laboratory
- The request is first received by the BSO, who has to validate it. By approving access, the BSO confirms that the person has received appropriate training specific to the requirements of the BSL2 laboratory in question.
- After approval by the BSO, the request is forwarded to the unit’s Principal Investigator (PI) or their designated representative.
- Following the PI approval, the request arrives to the biosafety team for final validation.
Bachelor students will not get access to the BSL 2 lab (they can access the BSL 2 lab only if accompanied by a trained and experienced person) and master students can get only partial access rights (Mon-Fri, 7h00 am to 7h00 pm).
For shared laboratories, the BSO and the PI have to confirm only requests coming from their own unit.
The maximum validity period for a BSL 2 access has been fixed to two years. The BSO will receive an alert indicating that an access is going to expire. The BSO will then decide to renew or not the access.
The access request for a BSL 2 laboratory is possible if:
- Future users have followed the FOBS3 mandatory training
- Future users are registered to the FOBS 3. In this case they will get only partial access (Mon-Fri, 7h00 am to 7h00 pm) until the training is completed.
A risk group 3 organism is considered by the World Health Organization as a “pathogen that usually causes serious human or animal disease but does not ordinarily spread from one infected individual to another (high individual risk, low community risk).” Effective treatment and preventive measures are usually available.
The biosafety level 3 (BSL3) laboratory is designed and provided for work with risk group 3 microorganisms. BSL3 containment requires the strengthening of the operational and safety programs over and above those for basic laboratories (BSL 1 and BSL 2).
For any support or additional information regarding Biosafety Level 3 activities, contact us.
Please contact the biosafety team at [email protected] and register for FOBS 3 training.
The Human Research Act (HRA, 810.30) requires that any research involving humans must be evaluated and authorized by the cantonal ethics commission. The collection of human blood samples (or other human samples) for experimental purposes falls into the category of projects that need to be approved by this commission.
The process has to be done on the CER-VD website.
How to work with human samples?
For any biosafety-related questions, such as where to work, which equipment to use, or how to handle waste, please don’t hesitate to contact us.
An import certificate from authorities might be necessary to import pathogens.
- An import certificate is not legally required to import of the majority of human pathogens.
- More information on the website of the Office of Public Health (FOPH).
- The Veterinary Office (FSVO) requires an import authorization for animal pathogens and for products of animal origin (e.g. food products).
- In order to get an import permit, it is necessary to fill the form on the FSVO website.
For BSL 2 biohazard spills:

- Inform your colleagues and make everyone leave the room
- Put on the door the biohazard spill alert notice
- Call 115
Only approuved biocides have to be used as decontaminants:
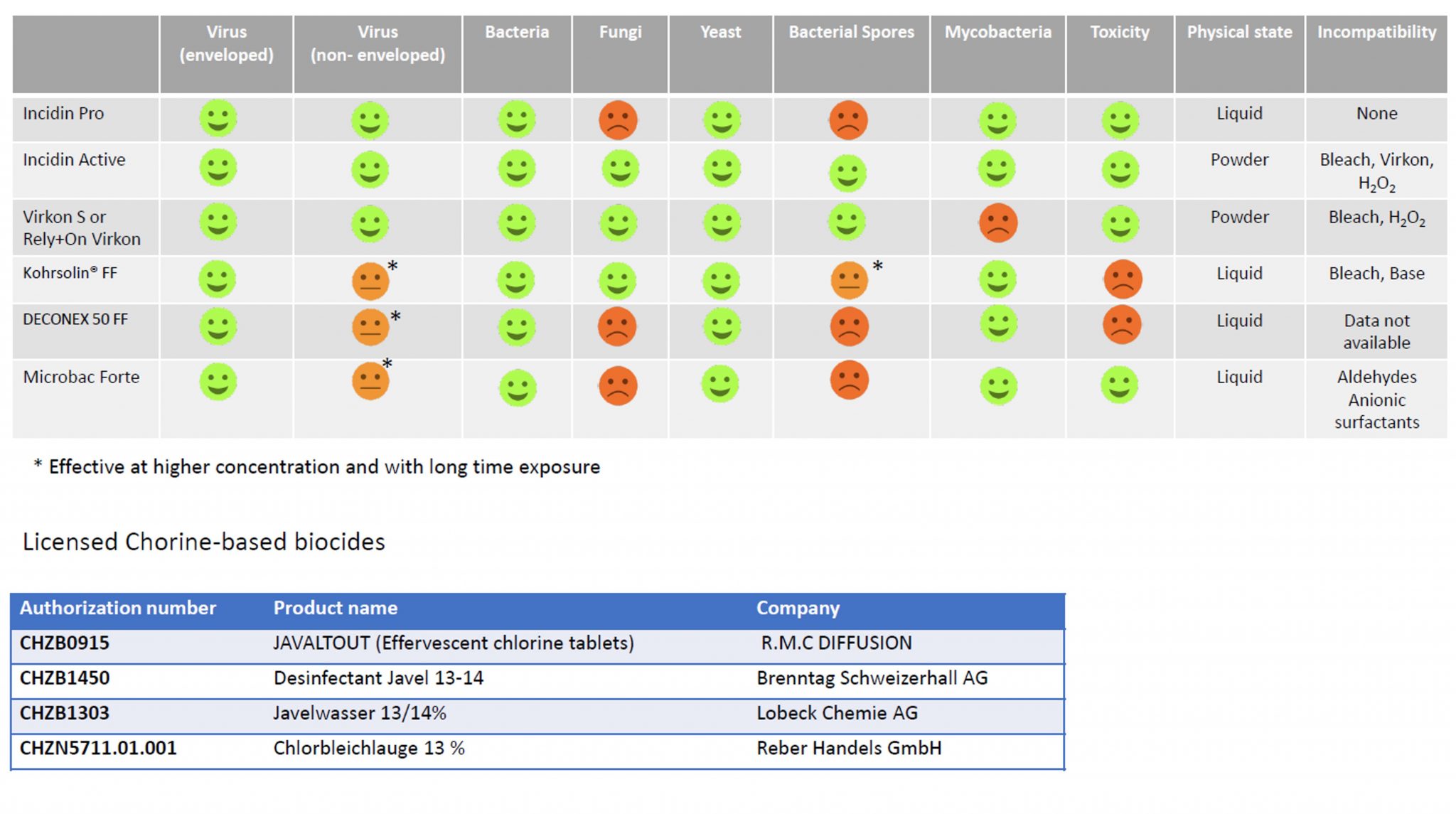
For the management of BSL 3 spills, please refer to the BSL 3 guidelines.
The Principal Investigator (PI), in collaboration with the BSO, is responsible for providing the biosafety team with the necessary information for drafting notifications and authorization documents.
This information typically includes:
- Title of the study
- Name and family name of BSO
- Name and family name of the contact person (who is typically the PI)
- Room number of all premises where biological activities are conducted
- Project description (scientific purpose, type of experiments), maximum half A4 page
- Maximum volume of liquid cultures
- Information on inactivation of solid waste (special transport or autoclave) and on liquid waste inactivation (autoclave or biocide).
- Name of organisms to be used (genus, species and strains). Also indicate whether these organisms will be genetically modified. In the case of GMOs, information on the type of genetic modification must be given (an article describing how organisms were modified is also ok)