Context
Biological smooth muscle is one of the three muscle tissue types found in the human body (in addition to cardiac and skeletal muscle) and are usually found in hollow organs such as the stomach or intestines. Tools capable of characterizing the function of smooth muscle cells i.e. contractile force and strain, are necessary for toxicology screening and the development of new drugs treating diseases such as asthma and functional bowel disease. Attempts made at developing such tools, though largely successful, are optical in nature and are therefore non-optimized for parallel, high-throughput and real-time measurement.
Towards High-throughput Cell Force Measurements
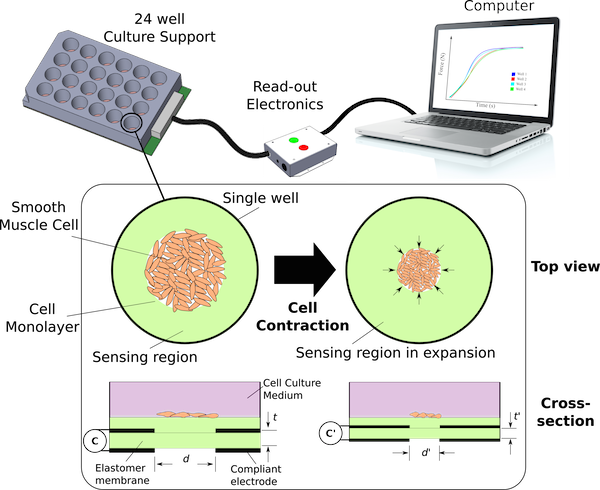
The LMTS, in collaboration with CSEM under the nano-tera.ch project “Breathe”, are developing a novel approach to measuring cell contraction force using of thin-film dielectric elastomer capacitive sensors. Smooth muscles cells cultured on the surface of a thin elastomer membrane sandwiched between two electrodes (cross-section shown above) induce deformations in the membrane as they contract. This contraction is detected electrically by the change in capacitance of the elastomer membrane, and the cell contractile force determined knowing the mechanical properties of the sensor.
The advantage of our approach is that the cell force is determined electrically and our capabilities for large area device fabrication (link to fabrication page) enables up-scaling of our system to have many (> 20) devices working in parallel for high-throughput measurement. Our models and initial cell characterizations indicate that a strain resolution of at least 1% is needed in order to detect cell contractions (for a silicone elastomer Young’s modulus ~ 0.1 MPa and membrane thickness < 20 µm).
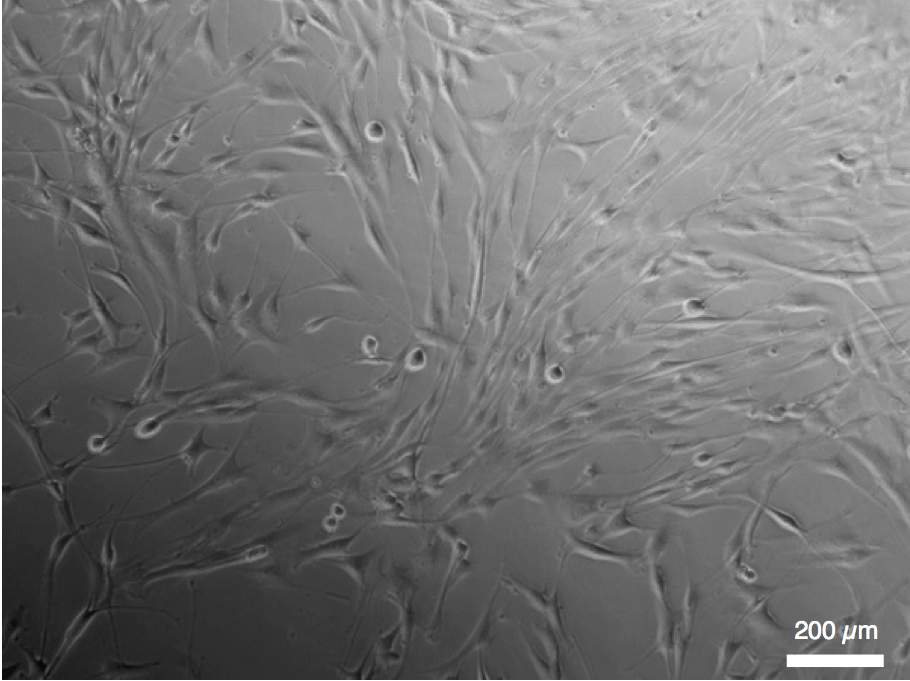
 Mircoscope images of human bronchial smooth muscle cells cultured on our thin elastomer membranes (left) and stretchable PDMS-carbon electrodes (right)
Mircoscope images of human bronchial smooth muscle cells cultured on our thin elastomer membranes (left) and stretchable PDMS-carbon electrodes (right)
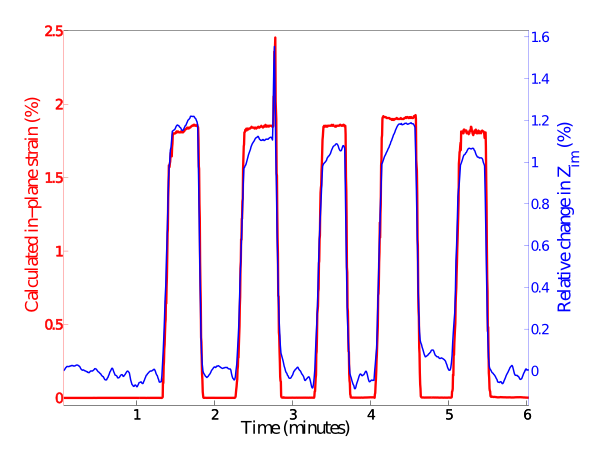
We’ve successfully built prototypes devices using biocompattible materials. Our initial results with these devices, shown below, show that we are able to achieve good strain resolution. In-plane strains of the order 1% were induced our prototype device shown above (simulating cell contraction) and produced a measurable, proportional and fast response with good signal to noise ratio.

Our EAPAD 2015 conference proceeding on this work can be found here.
| In collaboration with: | This project is funded by: |
 |
 |