Modification of cysteine
The high reactivity of EBX reagents with thiols make them well-suited for the functionalization of cysteine-containing biomolecules. In collaboration with the Adibekian group at the University of Geneva, an azide substituted EBX reagent could be used for the functionalization of proteins, even in living cells. The method was used for the proteome-wide profiling of targets of cysteine-reactive small molecules, which allowed the identification of one of the targets of curcumin, a natural product with high potential as anti-cancer agent (Angew. Chem., Int. Ed. 2015, 54, 10852. DOI: 10.1002/anie.201505641).
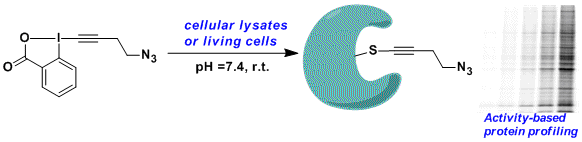
Potential applications of the thiol alkynylation reaction are not limited to proteins. Indeed, the Matile group at the University of Geneva, also used the same reagent for the termination of cell-penetrating poly(disulfide)s (Polym. Chem. 2016, 7, 3465. DOI: 10.1039/C6PY00562D ). In 2021, the Matile group also demonstrated that EBX reagents can be excellent inhibitors of thiol-mediated uptake across the cell membrane (Helv. Chim. Acta 2021, 104, e2100085. 10.1002/hlca.202100085).
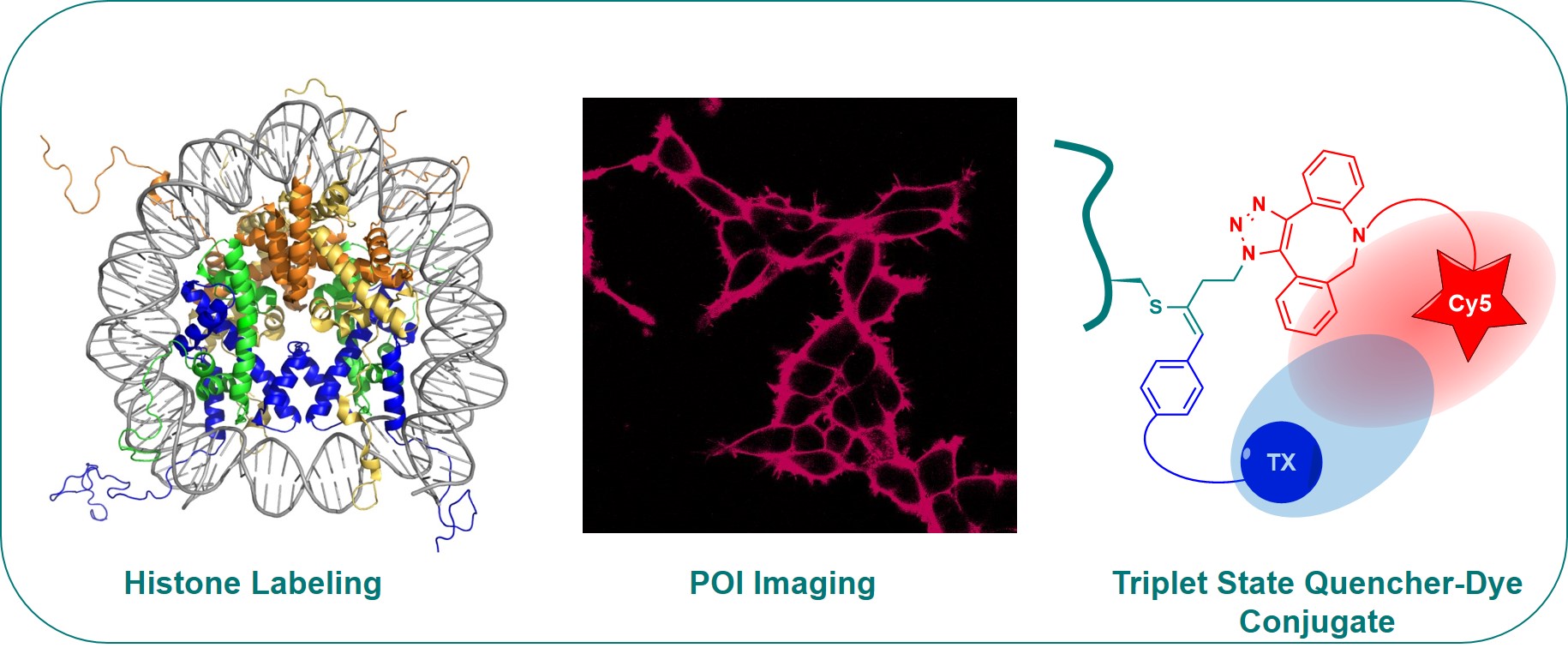
In our group, we wondered in particular if a general modification of biomolecules not limited to proteins with hyperreactive cysteines could be developed. In 2019, we finally succeeded in a general modification of cysteine and peptide in standard buffer solutions. However, the product obtained was not the alkynes, but the simple addition product of the thiol on EBX, probably due to the highly protic nature of the reaction media. The obtained conjugates were surprisingly stable to acids, bases and heating, and the reaction was successful from amino acids to histone proteins. Protein modification was done in collaboration with the Fierz group at EPFL. Particularly interesting was the use of azidated EBX reagents, which resulted in bioconjugate that are “doubly bioorthogonal”. A dye could be installed on the azide group, and a new palladium-catalyzed cross-coupling was developed to modify the hypervalent iodine bond. For example, both a dye and a triplet quencher were instal on the bioactive peptide substance P, allowing to measure single-molecule fluorescence of the peptide bound to cell receptors with enhanced life times (Chem 2019, 5, 2243-2263. DOI:10.1016/j.chempr.2019.06.022 ).
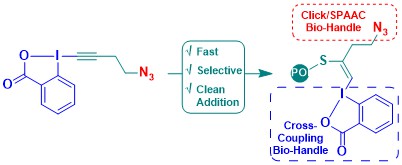
The development with alkyl substituted EBX reagents was highly interesting, but it was still difficult in several cases to control the alkyne to addition product ratio. Furthermore, the internal alkyne formed in these transformations was not well-suited for further transformations. In 2020, we then use TMS-EBX reagents for the direct introduction of terminal alkynes. In water buffer, the TMS group is first removed, and the obtained hypervalent iodine intermediate then converted to the terminal alkyne. The method was used in collaboration with the Wagner/Chaubet group for the functionalization of antibodies, and with the Adibekian group for proteomics. The use of this approach allowed to identify more proteins in the proteomic experiment compared to our first method (Angew. Chem., Int. Ed. 2020, 59, 10961-10970. (DOI:10.1002/anie.202002626 ).
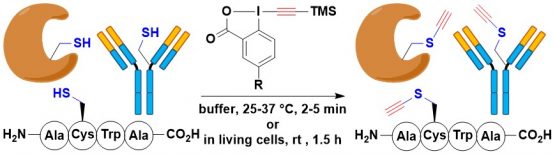
A limitation of EBX reagents for biomolecule functionalization was their low solubility in water. If the reagent is relatively small or polar, the solubility is sufficient to observe reactivity even in pure water buffer. However, the transfer of large lipophilic groups was not possible. This was an important limitation, as the lipophilization of peptides and proteins is an important process. The development of water-soluble EBX reagents has therefore been a long standing project at LCSO. In 2021, after four years of efforts, we succeeded in the synthesis and use of sulfate-substituted amphiphilic EBX reagents, which could be used in pure buffer for the functionalization of cysteine in peptides and proteins. Interestingly, in this case the alkyne was the major product. Under acidic conditions, the thioalkynes could be converted into thioesters, which can be cleaved under basic conditions (Angew. Chem., Int. Ed. 2021, 60, 17963-17968. (10.1002/anie.202106458).
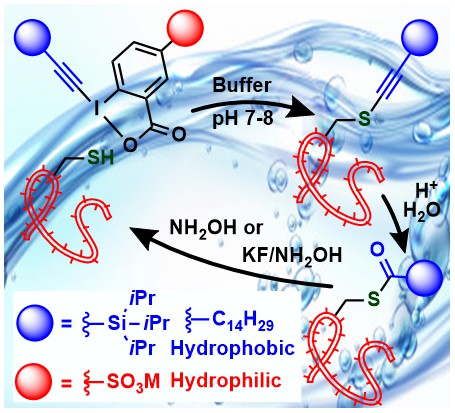
Peptide stapling is an important strategy for stabilizing peptides, especially alpha-helices, and improve their affinity to the targets. When considering the exceptional reactivity of EBX reagents with thiols, we speculated that they would be ideally suited for peptide stapling using cysteine functionalization in unprotected peptides. We therefore developed two new types of reagents: Bis-EBX compounds for cysteine-cysteine stapling, and EBX-activated ester reagents for cysteine-lysine stapling. Both reagents could be used for i+4 and i+7 peptide stapling, with the cysteine-lysine stapling reagents being especially general and efficient. The thioalkyne incorporated in the tether could be directly used in a ruthenium-catalyzed cycloaddition with azides. In collaboration with the Heinis group we demonstrated that one of the obtained stapled peptide indeed showed increase affinity for the protein MDM2 (Angew. Chem., Int. Ed. 2021, 60, 9022-9031. (DOI:10.1002/anie.202014511 ).
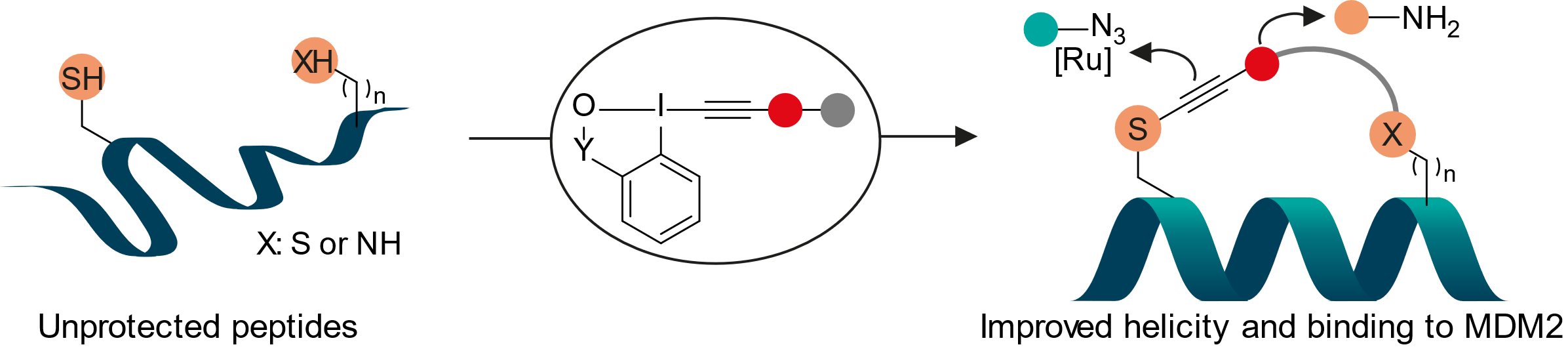
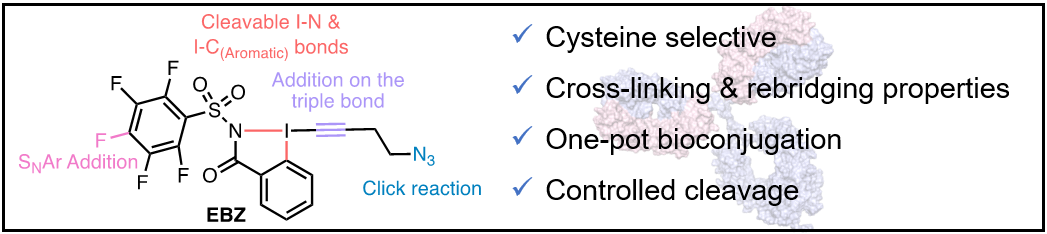
We became also interested in using nitrogen-based EBZ reagents for biomolecule functionalization. In fact, the substituent on nitrogen allows the introduction of a further handle for bioconjugation. In 2023, we introduced a new reagent with a polyfluorinated arylated sulfonamide on the nitrogen. This reagent enabled the cross-coupling of two different thiols based on the different rates of addition of thiols onto the EBZ and nucleophilic aromatic substitution. Together with the Chaubet group at the university of Strasbourg, we used this reagent for antibody rebridging and functionalization (Chem. Eur. J. 2023, 29, e202302689. 10.1002/chem.202302689).
Modification of tyrosine
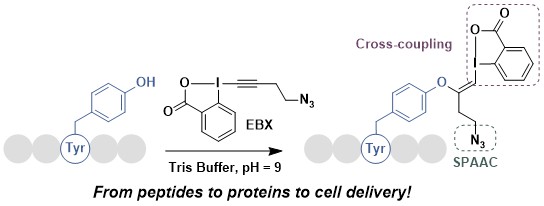
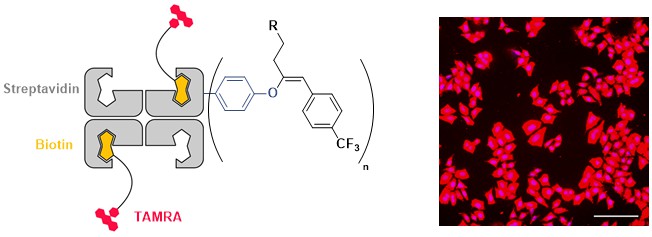
From our work on small molecules, we knew that EBX reagents can also react with phenols to give the VBX addition products. However, the reactivity was much lower than for thiols, so it was not clear if this transformation could work under physiological conditions. In 2022, we were indeed able to develop the first tyrosine selective functionalization of peptides and proteins using EBX reagents. The reaction is much slower than with thiols (1 day instead of 1 min), but other nucleophilic amino acids still remain untouched. In collaboration with the Matile group at the University of Geneva, we used this method to modify streptavidin to enable it to pass the cell membrane. We also collaborated with the Stallforth group at the University of Jena for the functionalization of the natural lipopetide jagaricin (Chem. Sci. 2022, 13, 12808. DOI: 10.1039/D2SC04558C).
Decarboxylative functionalization of peptides
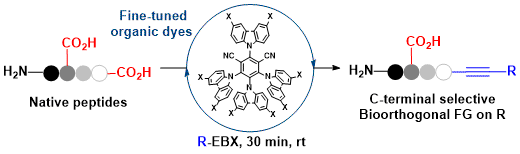
As a second approach towards biomolecule functionalization, we wondered if our decarboxylative alkynylation method based on photoredox catalysis could be extended to peptides. This was challenging as the conditions we originally developed did not work on peptides. Fortunately, using CzIPN as catalyst and aryl-EBX reagents, we were able to develop a highly efficient decarboxylative alkynylation of peptides. Investigation on tetrapeptides demonstrated that the reaction tolerated most amino acids, with the exception of tryptophan. In case of cysteine, double alkynylation was observed. The reaction was selective for the C-terminus, and no alkynylation of carboxylic acid side-chains was observed. This was an important step towards the dream of a decarboxylative alkynylation of the C-terminus of proteins, but major hurdles still need to be overcome, as the efficiency of the reaction diminishes drastically with increased length of the peptide or amount of water. ( Angew. Chem. Int. Ed. 2019, 58, 8182-8186. DOI:10.1002/anie.201901922 )
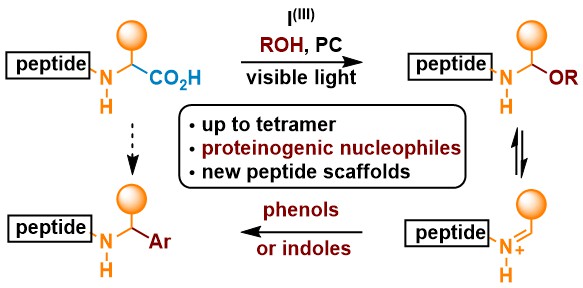
When using simple benziodoxole acetate as oxidant, we found out that it was possible to perform a two-electron oxidation under photoredox conditions to give highly reactive iminium intermediates. A well-established ruthenium photocatalyst gave the best result in this case. Alcohols reacted fast with the iminium intermediate to give isolable N,O-acetals. Alternatively, Friedl-Crafts reactions with both phenols and indoles were possible to give new non-natural peptides. When peptidic nucleophiles were used in the reaction, new types of peptides with non natural connections could be obtained. (Chem. Sci. 2021, 12, 2467-2473. DOI:10.1039/D0SC06180H )
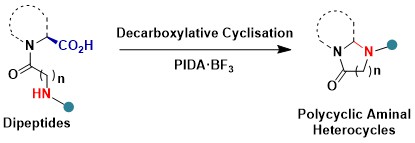
In absence of external nucleophiles, we discovered that a cyclization on the amide nitrogen of the neighboring peptide can occur. Interestingly, this reaction is best done by PIDA activated with boron trifluoride. This decarboxylative cyclization gives access to various polycyclic aminal heterocycles (Chem. Commun. 2022, 58, 3473. DOI:10.1039/D2CC00167E )
Azidation of peptides
Since ten years our group has been working on azidation using hypervalent iodine reagents. The azidation of peptides and proteins is therefore also a topic of high interest to complement alkyne and hypervalent iodine chemistry. In 2022, we published a first example of azidation of peptides, which proceeds selectively on the C-H bond of carbamate protected proline. In this case, a safer protocol can be used, in which the active hypervalent iodine reagent is directly generated in situ through oxidation with meta-perchlorobenzoic acid. (Chem. Eur. J. 2022, 28, e202200368. DOI:10.1002/chem.202200368).
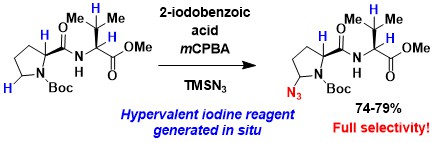
Peptides hypervalent iodine reagents
Over the years, we had discovered the unique properties of hypervaletn iodine reagents for the modification of peptides and proteins. Since 2021, we started to investigate another approach: the installation of EBX reagents unto peptides for their biorthogonal functionalization. In 2023, our efforts culminated in the first synthesis of such Peptide-EBX reagents via the functionalization of either lysine or the N-Terminus. The reagents could be conjugated with thiols, and especially enabled the first photoredox-mediated alkynylative macrocyclization of peptides (Angew. Chem., Int. Ed. 2023, 62, e202306036. DOI: 10.1002/anie.202306036).
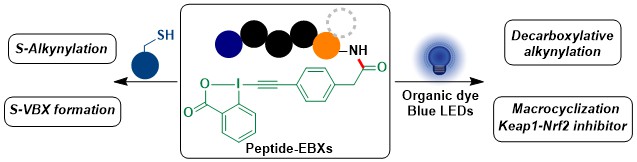
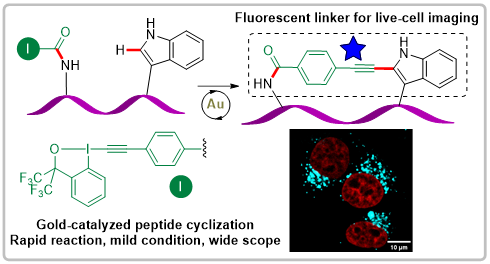
We have now just started to investigate applications of these new peptide-EBX reagents. The next step was to re-examine the potential of the gold-catalyzed alkynylation of indoles with EBX reagents in the much more complex setting of peptide macrocyclization. This reaction in fact enabled efficient coupling between EBx reagents and tryptophan for accessing a broad scope of macrocycles. Using the more stable bistrifluoromethylated reagents in this case facilitated greatly the synthesis and purification of the peptide-EBxs. An advantage of this method is that the synthesized macrocycles are themselves fluorescent and can be used for cell imaging (J. Am. Chem. Soc. 2023, 145, 26525-26531. 10.1021/jacs.3c09261).
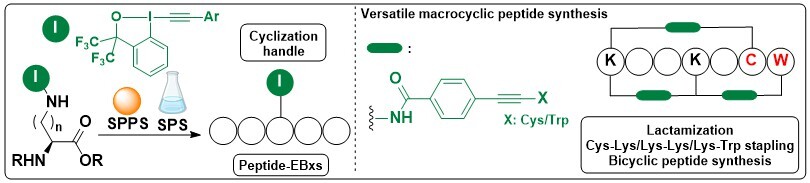
Using our original methodology, it was difficult to functionalize selectively peptides containing multiple lysines, and multi-step orthogonal protecting group strategies were needed. Therefore, another approach was attempted, in which the hypervalent iodine reagent was introduced since the start into a non-natural amino acid. This approach was challenging, as the reactive hypervalent bond would need to survive multiple coupling/deprotection steps. In 2024, we demonstrated that bistrifluoromethyl-derived Bx reagents were in fact suitable. The building blocks derived from standard protected amine-side chain amino acids could be used both in standard solution and solid phase peptide synthesis. The diverse methodologies developed in the group could then be used to cyclize and/or functionalize the obtained peptide-EBxs. With this breakthrough, the methodology can now be used by any laboratory equipped with a peptide synthesizer, as we will be happy to send the required non-natural amino acids! (Angew. Chem., Int. Ed. 2024, 63, e2202404747. 10.1002/anie.202404747).
Site-Selective Protein Modification
The capacity of installing EBX reagents on peptides allowed us also to address a long-standing challenge in protein functionalization: Site-selective modification. By using a directing peptide as ligand, we aimed to functionalize only one cysteine in the KELCH domain of the protein KEAP1, which contains 8 free cysteines. For this purpose, we designed new peptide-EBX reagents with the peptides bound on the arene core. In 2025, we achieved our goal and could develop a highly selective modification of cysteine 434, which is important for the KEAP1-NRF2 interaction. Using silyl-substituted EBXs gave access to ligand free alkyne products, whereas the us of alkyl- or aryl-substituted EBXs allowed the formation of covalent VBX adducts. (J. Am. Chem. Soc. 2025, 147, 42524-42531. DOI: 10.1021/jacs.5c13391). These proof of concept results open the door for the modification of other residues and targets.

Functionalization of peptides and proteins with hypervalent iodine reagents: An emerging area
The results obtained in our group since 2015 for the functionalization of peptides and proteins with EBX reagents are highly promising. Other research groups have also used hypervalent iodine reagents for biomolecule functionalization. The potential for further applications is enormous, when considering the unique reactivity of hypervalent iodine reagents, combined with their stability in water and lower toxicity in comparison with metal reagents. This motivated us to write a review about this emerging area of research in 2021 ( Angew. Chem. Int. Ed. 2021, 60, e202112287. DOI:10.1002/anie.202112287).
As the area was progressing very fast, we published in 2025 an update on the functionalization of peptides and proteins as well as macrocyclization methods using hypervalent iodine reagents, this time focusing more on the results obtained in our group (Acc. Chem. Res.2025, 58, 2852-2861. DOI:10.1021/acs.accounts.5c00451).

Peptides functionalization without hypervalent iodine reagents
Non natural amino acids
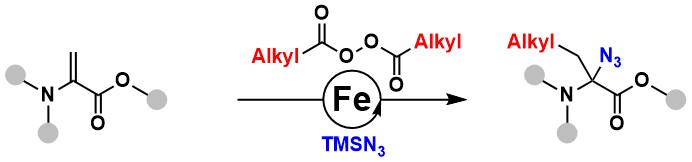
Although the focus of our group is on the use of hypervalent iodine reagents, we naturally do not hesitate to use other methodologies when they are not needed. In 2023, our interest in azidation led us to develop a new iron-catalyzed carboazidation of dehydroalanines. In this case, the use of peroxides as alkyl radical sources and trimethylsilyl azide worked best. This method gave access to unprecedented alpha-azidated amino acid building blocks, which could be used for synthesizing new aminal based peptide derivatives (Org. Lett. 2023, 25, 6791-6795. DOI: 10.1021/acs.orglett.3c02153).
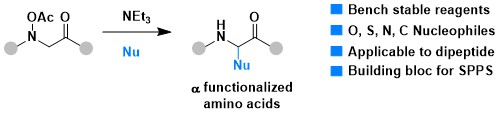
In 2024, we also developed a new approach for accessing alpha-functionalized amino acids based on an interrupted Polonovski strategy. This approach complements existing methods focusing on C-H functionalization usually requiring more easily oxidizable amines, such as anilines (Org. Lett. 2024, 26, 456-460. 10.1021/acs.orglett.3c03603).
Peptodyes
In 2025, we serendipitously discovered the decarboxylative C-terminal substitution of cyanobenzene dyes with peptides. We then optimized this transformation and were able to functionalize a broad range of peptides with cyanobenzenes. As this first generation of peptodyes were obtained as a mixture of diastereoisomers, we then developed a second strategy for accessing peptodyes bases on the nucleophilic aromatic substitution of cysteine on fluorinated cyanobenzenes. Both types of peptodyes were competent photocatalysts in biological medias for decarboxylative alkynylation and thiol ene reactions. As a proof of concept for a first application, we then developed a proximity-driven modification of integrin based on the generation of reactive nitrene intermediates by a peptodyes bearing an integrin binder. In principle, this methodology can be used for the labelling of any target once a peptide binder is available (Angew. Chem., Int. Ed. 2025, 64, e202507602. 10.1002/anie.202507602).

If you are working in chemical biology and would like to try some of our compounds, don’t hesitate to contact us: we are always eager to share reagents to attempt new applications!