The SeleCHEM project (Overcoming the Selectivity Challenge in Chemistry and Chemical Biology via Innovative Tethering Strategies) had the very ambitious goal to develop selective processes in synthetic chemistry. One half of the project is focusing on catalysis and small molecules modification, and the other half on biomolecule functionalization. It combines two of the most successful research areas of the group: tether chemistry and biomolecule functionalization. You can find below the publications related to this project.
Journal Articles
2025
Peptide-Carbazolyl Cyanobenzene Conjugates: Enabling Biomolecule Functionalization via Photoredox and Energy Transfer Catalysis
Xing-Yu Liu, Wei Cai, Anne-Sophie Chauvin, Beat Fierz and Jerome Waser
Angew. Chem., Int. Ed. 2025, 64, e202507602. (10.1002/anie.202507602) Open Access! Green Access Raw data

2024
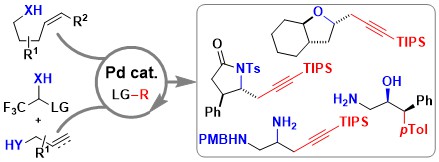
Enamine Synthesis via Regiocontrolled 6-endo-dig and 5-exo-dig Tethered Carboamination of Propargylic Alcohols
Helena Solé-Àvila, Mikus Puriņš, Lucas Eichenberger and Jerome Waser
Angew. Chem. Int. Ed. 2024, 63, e202411383. (10.1002/anie.202411383) Open Access! Raw data
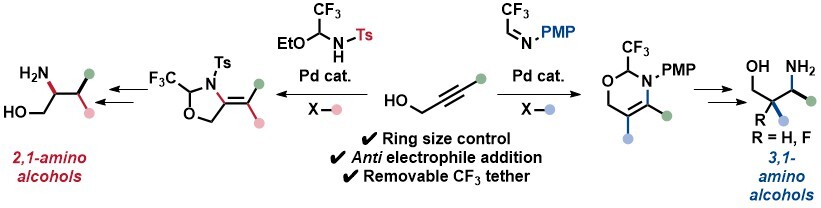
Hypervalent Iodine Amino Acid Building Blocks for Bioorthogonal Peptide Macrocyclization
Xing-Yu Liu, Olha Mykhailenko, Adriana Faraone and Jerome Waser
Angew. Chem. Int. Ed. 2024, 63, e202404747. (10.1002/anie.202404747) Open Access! Raw data
2023
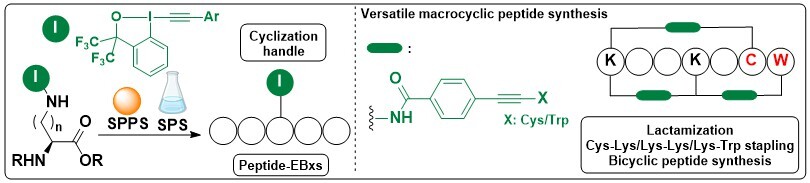
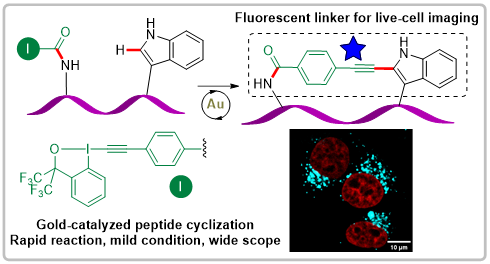
Synthesis of Fluorescent Cyclic Peptides via Gold(I)-Catalyzed Macrocyclization
Xing-Yu Liu, Wei Cai, Nathan Ronceray, Aleksandra Radenovic, Beat Fierz and Jerome Waser
J. Am. Chem. Soc. 2023, 145, 26525-26531. (10.1021/jacs.3c09261) Open Access! Raw data
One-pot synthesis of functionalized bis(trifluoromethylated)benziodoxoles from iodine(i) precursors
Tobias M. Milzarek, Nieves P. Ramirez, Xing-Yu Liu and Jerome Waser
Chem. Commun. 2023, 59, 12637-12640. (10.1039/D3CC04525K) Green Access Raw data
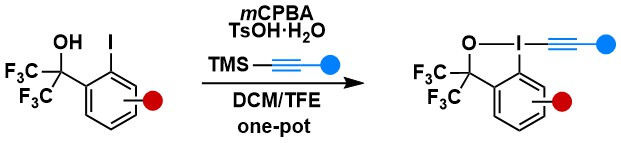
Cysteine-Cysteine Cross-Conjugation of both Peptides and Proteins with a Bifunctional Hypervalent Iodine-Electrophilic Reagent
Ilias Koutsopetras, Abhaya Kumar Mishra, Rania Benazza, Oscar Hernandez-Alba, Sarah Cianférani, Guilhem Chaubet, Stefano Nicolai and Jerome Waser
Chem. Eur. J. 2023, 29, e202302689. (10.1002/chem.202302689) Open Access!
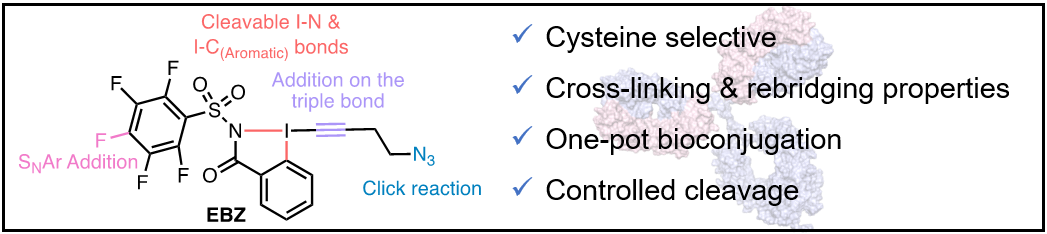
Synthesis of Trifluoromethylated Alkenes: Hypervalent Iodine Meets High-Valent Copper
Tobias Michael Milzarek and Jerome Waser
Angew. Chem., Int. Ed. 2023, e202306128. (DOI: 10.1002/anie.202306128) Open Access! Raw data
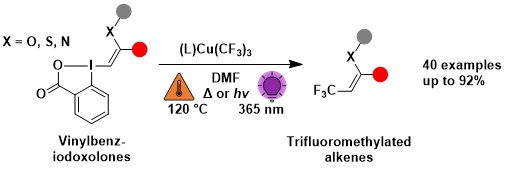
Peptide-Hypervalent Iodine Reagent Chimeras: Enabling Peptide Functionalization and Macrocyclization
Xing-Yu Liu, Xinjian Ji, Christian Heinis and Jerome Waser
Angew. Chem., Int. Ed. 2023, e202306036. (DOI: 10.1002/anie.202306036) Open Access! Raw data
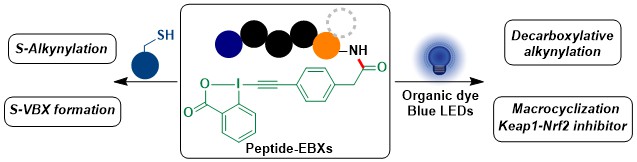
Synthesis of propargyl silanes from terminal alkynes via a migratory Sonogashira reaction
Mikus Purins, Lucas Eichenberger, and Jerome Waser
Chem. Commun. 2023, 59, 7931-7934. (DOI: 10.1039/D3CC01847D ) Open Access! Raw data
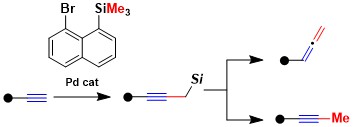
2022
Pd(II)-Catalyzed Aminoacetoxylation of Alkenes Via Tether Formation
Thomas Rossolini, Ashis Das, Stefano Nicolai and Jerome Waser
Org. Lett. 2022, 24, 5068-5072. (10.1021/acs.orglett.2c01838) Raw data
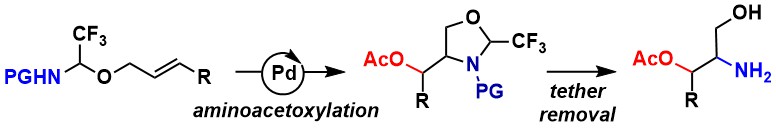
Palladium-Catalyzed trans-Hydroalkoxylation: Counterintuitive Use of an Aryl Iodide Additive to Promote C–H Bond Formation
Ashis Das, Luca Buzzetti, Mikus Puriņš and Jerome Waser
ACS catal. 2022, 12, 7565-7570. (10.1021/acscatal.2c01809) Raw data
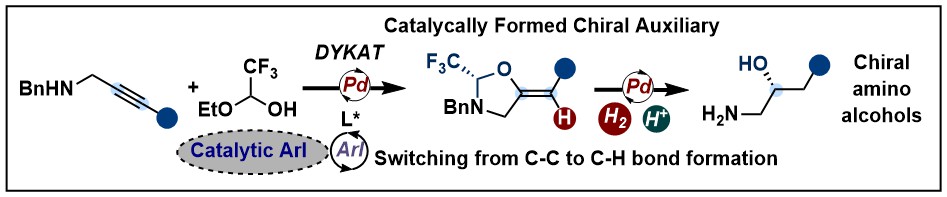
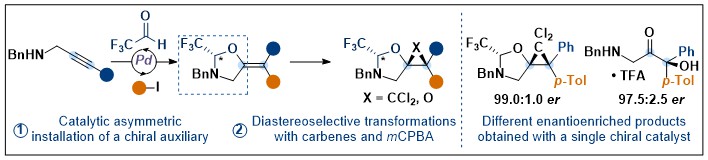
Asymmetric Cyclopropanation and Epoxidation via a Catalytically Formed Chiral Auxiliary
Mikus Purins and Jerome Waser
Angew. Chem., Int. Ed. 2022, 61, e202113925. (10.1002/anie.202113925) Raw data Open Access!
2021
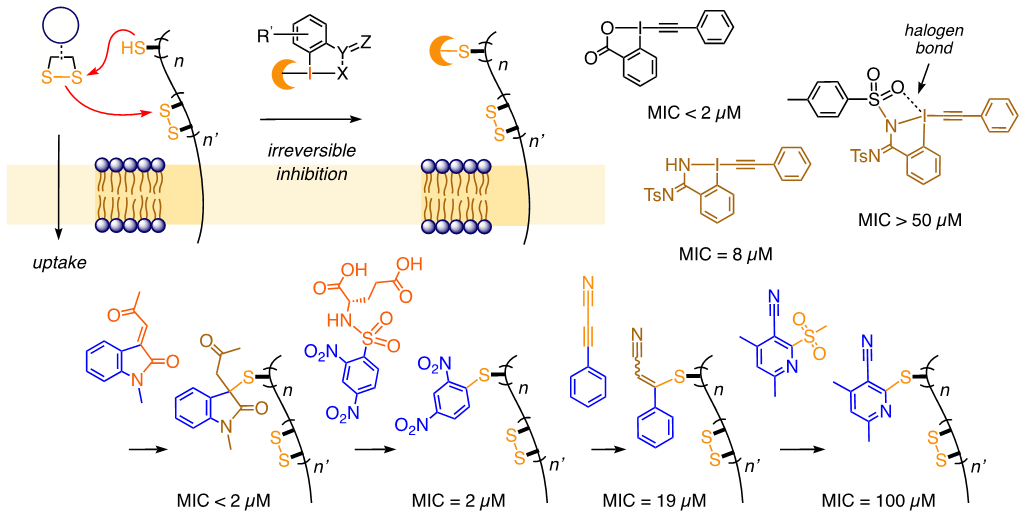
p style=”text-align: justify;”>Inhibition of Thiol-Mediated Uptake with Irreversible Covalent Inhibitors
Bumhee Lim, Yangyang Cheng, Takehiro Kato, Anh-Tuan Pham, Eliott Le Du, Abhaya Kumar Mishra, Elija Grinhagena, Dimitri Moreau, Naomi Sakai, Jerome Waser and Stefan Matile
Helv. Chim. Acta 2021, 104, e2100085. (10.1002/hlca.202100085) Open Access!
Collaboration: The biological screenings were performed at the Matile group .
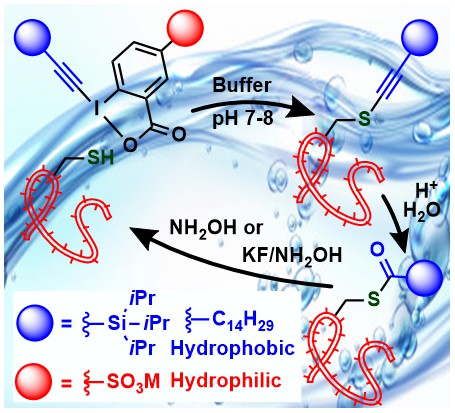
Amphiphilic Iodine(III) Reagents for the Lipophilization of Peptides in Water
Abhaya Kumar Mishra, Romain Tessier, Durga Prasad Hari and Jerome Waser
Angew. Chem., Int. Ed. 2021, 60, in press. (10.1002/anie.202106458) Open Access!
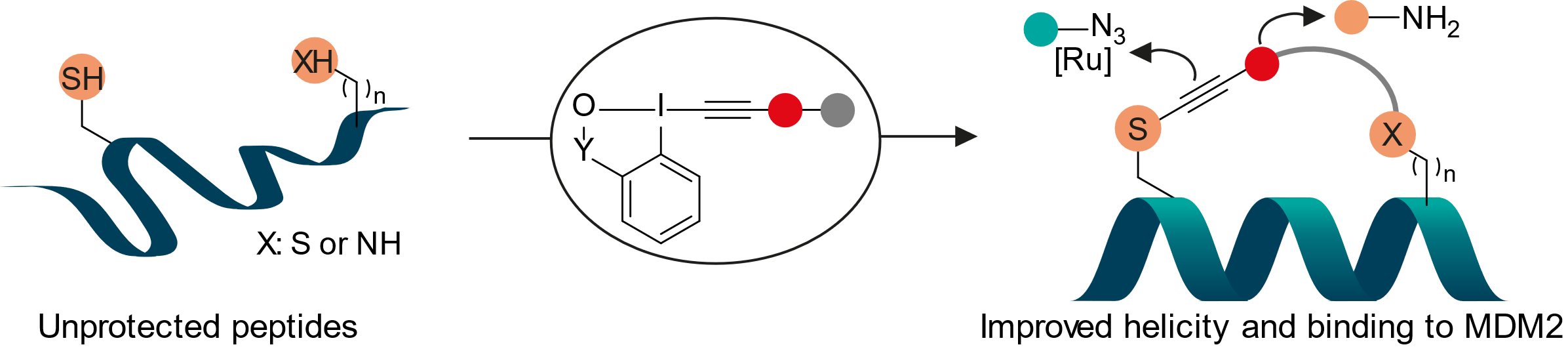
Cys‐Cys and Cys‐Lys Stapling of Unprotected Peptides Enabled by Hypervalent Iodine Reagents
Javier Ceballos, Elija Grinhagena, Gontran Sangouard, Christian Heinis and Jerome Waser
Angew. Chem., Int. Ed. 2021, 60, 9022-9031. (10.1002/anie.202014511) Open Access!
Collaboration: The binding affinity of the stapled peptides was measured at the Heinis group .
2020
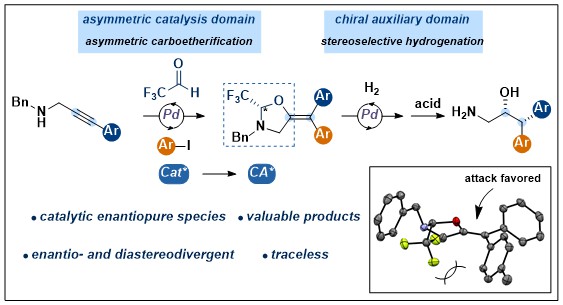
Enantioselective Carboetherification/Hydrogenation for the Synthesis of Amino Alcohols via a Catalytically Formed Chiral Auxiliary
Luca Buzzetti, Mikus Purins, Phillip D. G. Greenwood and Jerome Waser
J. Am. Chem. Soc. 2020, 142, 17334-17339. (10.1021/jacs.0c09177 ) Raw data Open Access!
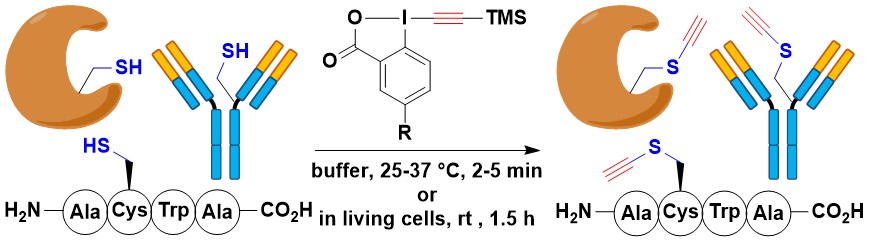
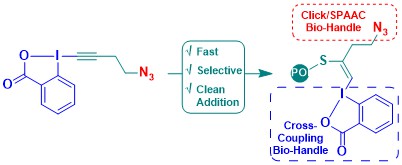
Ethynylation of Cysteines from Peptides to Proteins in Living Cells
Romain Tessier, Raj Kumar Nandi, Brendan G. Dwyer, Daniel Abegg, Charlotte Sornay, Javier Ceballos, Stephane Erb, Sarah Cianferani, Alain Wagner, Guilhem Chaubet and Jerome Waser
Angew. Chem., Int. Ed. 2020, 59, 10961-10970. (10.1002/anie.202002626 ) Green Access
Collaboration: Antibody modification studies were done in the Wagner/Chaubet group and proteomic studies were done in the Adibekian group .
2019
”Doubly Orthogonal” Labeling of Peptides and Proteins
Romain Tessier, Javier Ceballos, Nora Guidotti, Raphael Simonet-Davin, Beat Fierz and Jerome Waser
Chem 2019, 5, 2243-2263. (DOI:10.1016/j.chempr.2019.06.022 ) Green Access
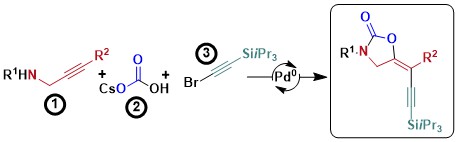
Palladium‐Catalyzed Carboxy‐Alkynylation of Propargylic Amines Using Carbonate Salts as Carbon Dioxide Source
Phillip D. G. Greenwood and Jerome Waser
Eur. J. Org. Chem. 2019, 2019, 5183-5186. (DOI:10.1002/ejoc.201900500 ) Green Access
Reviews
2022
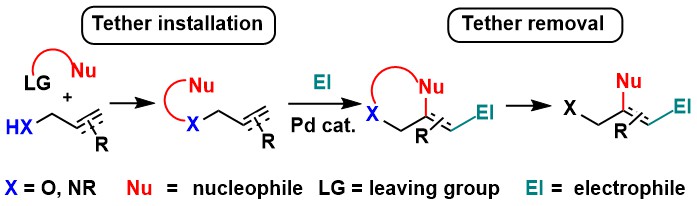
Pd-catalyzed functionalization of alkenes and alkynes using removable tethers
Ashis Das and Jerome Waser
Tetrahedron 2022, 128, 133135. (DOI:10.1016/j.tet.2022.133135) Open Access!
2021
Hypervalent Iodine-Mediated Late-Stage Peptide and Protein Functionalization
Emmanuelle M. D. Allouche, Elija Grinhagena and Jerome Waser
Angew. Chem. Int. Ed., in press. (DOI:10.1002/anie.202112287) Open Access!
Palladium-Catalyzed Functionalization of Olefins and Alkynes: From Oxyalkynylation to Tethered Dynamic Kinetic Asymmetric Transformations (DYKAT)
Stefano Nicolai, Ugo Orcel, Bastian Muriel, Phillip D. G. Greenwood, Luca Buzzetti, Mikus Purins and Jerome Waser
Synlett 2021, 32, 472-487. (DOI: 10.1055/a-1308-0021 ) Green Access