Benziodoxoles: A toolbox of reagents for organic synthesis
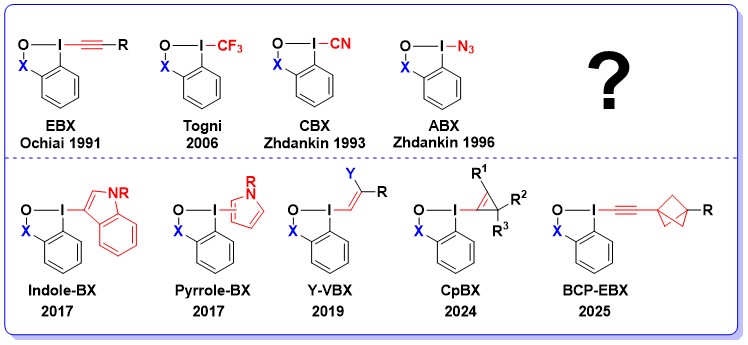
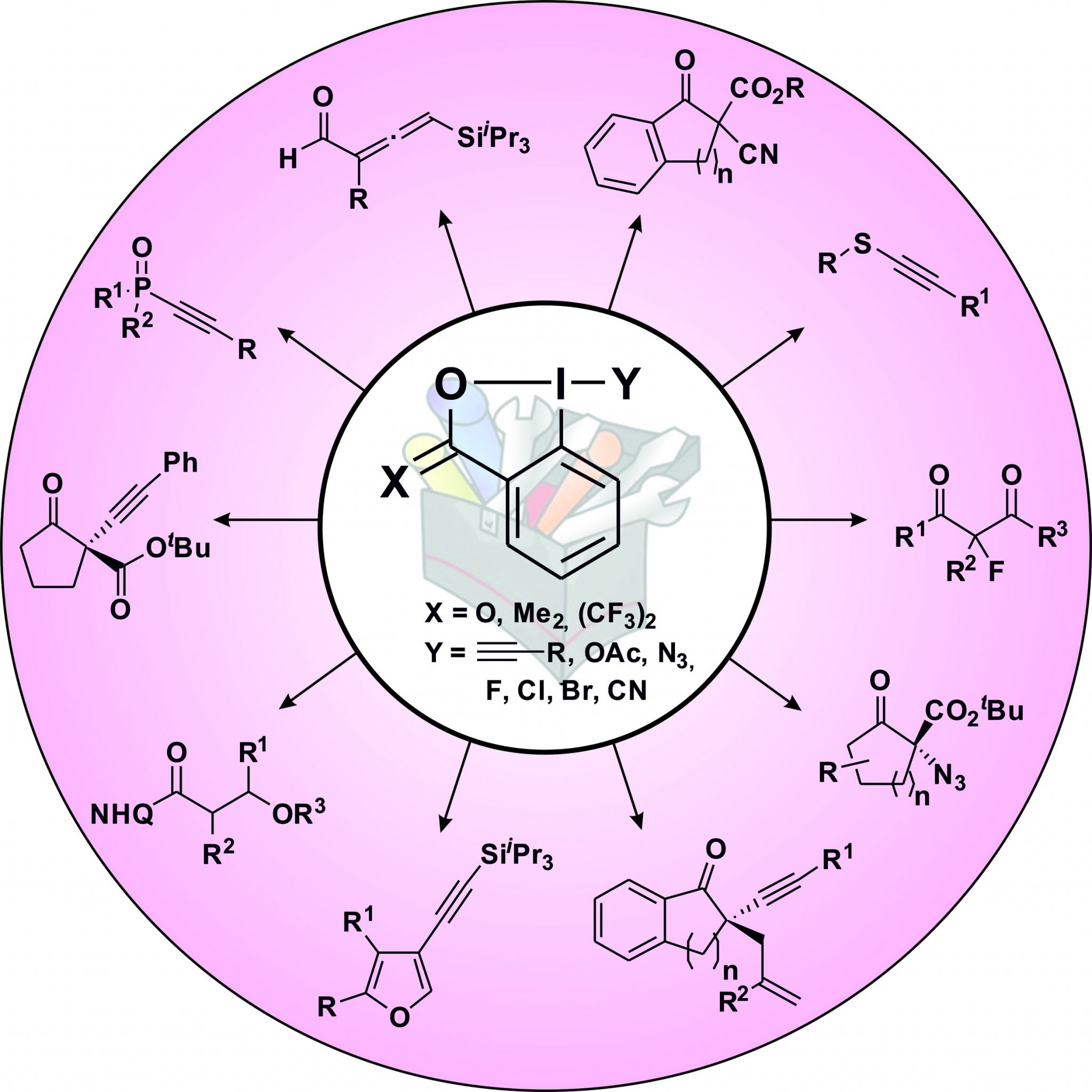
Up to twenty years ago, cyclic hypervalent iodine reagents, such as benziodoxoles, had been only scarcely employed for the introduction of new functional groups in organic chemistry. In fact, the most frequently used cyclic hypervalent iodine reagents were iodine (V) compounds, such as IBX and Dess-Martin periodinane in oxidation reactions. With our work on alkyne transfer with ethynylbenziodoxoles (EBXs) and the work of Togni and co-workers on trifluoromethylation, the exceptional properties of benziodoxoles reagents for atom-transfer reactions have become for the first time truly apparent (Chem. Commun. 2011, 47, 102. DOI:10.1039/C0CC02265) . It is our conviction that these properties will also be useful for the transfer of other functional groups. In view of a first step in this direction, we were particularly interested in the azido- and cyano- benziodoxole reagents, two further “treasures” first synthesized in the Zhdankin group. Indeed, cyanides and azides are among the most versatile functional groups in organic chemistry. Most methods used to introduced them in organic molecules are based on the nucleophilicity of the azide and cyanide ions, and more general and versatile electrophilic synthons are urgently needed. In 2017, our group further introduced Indole-BX and Pyrrole-BX as new reagents for the Umpolung of indoles and pyrroles. In 2019, we introduce a practical method for the synthesis of heteroatom-substituted VBX (vinylbenziodoxole) reagents. In 2024, the first access to Cyclopropenylbenziodoxoles (CpBxs) was disclosed. Finally, in 2025, we combined EBX reagents with bicyclopentanes for easy access to alkynylbenzene isosteres. Of course, the research for new, yet undisclosed reagents continues to be an important topic of research in our group, and both modifications of the benziodoxole core structure and the transferred functionality offer exciting opportunities for further development.
Azidation with azidobenziodoxolones
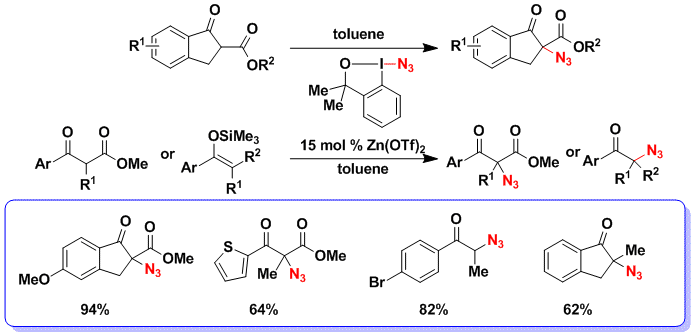
With azidobenziodoxole reagents, we discovered a fast and clean azidation of cyclic ketoesters without the addition of any additive (Org. Lett. 2013, 15, 3246. DOI:10.1021/ol401229v) . We preferred the use of the dimethyl reagent, as it was both more efficient and safer to use (smooth decomposition vs explosion for the carboxy reagent). Nevertheless, the azidation did not work in the case of non-cyclic ketoesters or ketones as substrates. In this case, we found that it was possible to further activate the reagent using zinc triflate as catalyst. Under these conditions, both non-cyclic ketoesters and silyl enol ethers could be azidated successfully.
Our next goal was to develop an azidation of non-activated olefins. In 2017, we developed two complementary approaches for the azidolactonization of olefins, using either photoredox or Lewis acid catalysis (Chem. Eur. J. 2017, 23, 9501. DOI:10.1002/chem.201702599) .
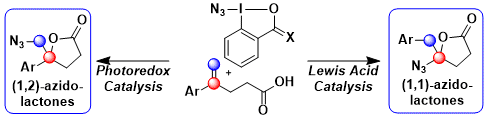
As Azidobenziodoxolone (ABX, Zhdankin reagent) is a stronger oxidant, it was an ideal reagent for the generation of azide radicals in presence of a copper photocatalyst and blue LED. The formed azide radical can then react with an olefin, generating a benzylic radical, followed by oxidation to the carbocation and attack of the carboxylic acid to give 1,2-azidolactones. Unfortunately, ABX is unstable and can explode spontaneously (see Stability Data). In contrast, Azidodimethylbenziodoxole (ADBX) could be activated by a Lewis acid to give the corresponding iodonium. Reaction wiht the olefin, followed by lactonization, 1,2-aryl shift and azidation gave then the observed 1,1-azidolactones.
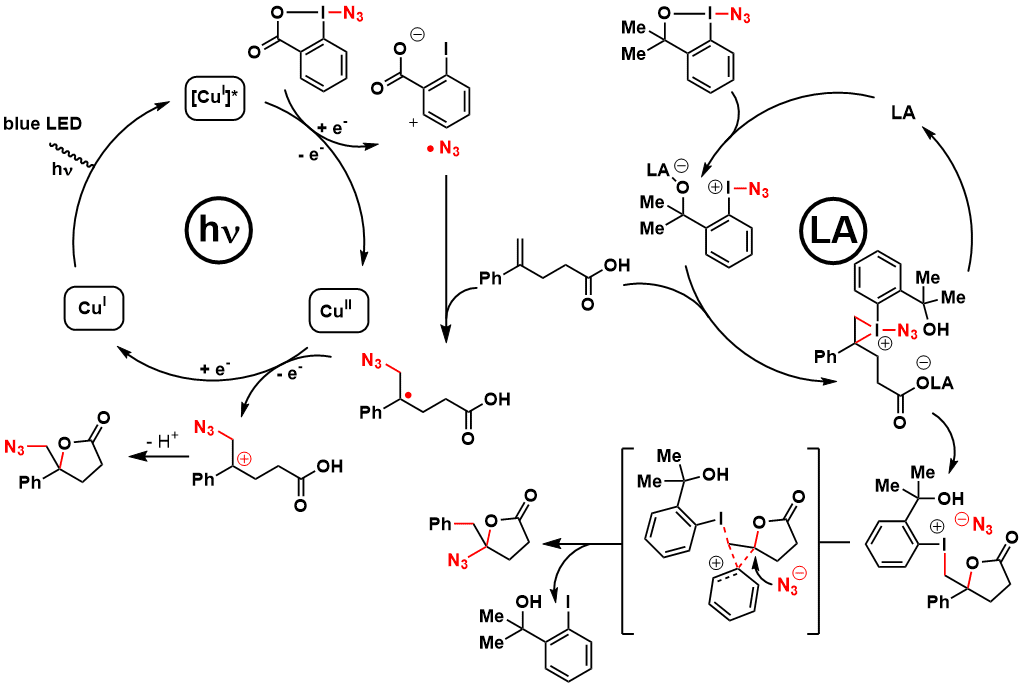
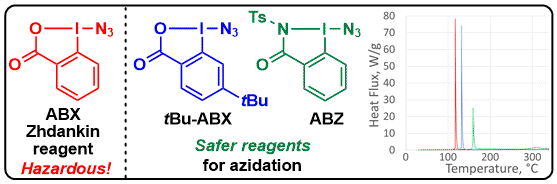
When working with a highly pure sample of Zhdankin reagent, an explosion occurred, leading to injury to the researcher. We therefore collaborated with the safety department at EPFL and TUV Sud AG in Basel to have a better idea of the safety profile of this reagent. The results were frightening, indicating very high shock and friction sensitivity. Fortunately, other derivatives were safer to use and displayed similar reactivity. The benziodazolone derivative (ABZ) was especially efficient for the generation of azido radicals under photoredox conditions (J. Org. Chem. 2018, 83, 12334-12356. DOI:10.1021/acs.joc.8b02068 ).
A long standing goal of the group was to develop efficient C-H azidation methods. Unfortunately, we were scooped by other groups (Hartwig, Stahl) in this area of research. Interestingly the Zhu group at EPFL was also developing such transformations. In 2024, we finally sumarized the finding of both our groups in this area, showing that in this case other oxidants and nucleophilic azide sources could be used directly (Helv. Chim. Acta 2024, 107, e202400004 10.1002/hlca.202400004).
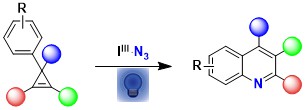
In 2021, we combined two fields of research in our group: azidation with hypervalent iodine reagents and small ring chemistry to develop a new synthesis of quinolines starting from cyclopropenes. In this work, direct activation of ABZ with light in presence of acetoxybenziodoxolone was sufficient to generate the required azido radical (Org. Lett. 2021, 23, 5435-5439. DOI:10.1021/acs.orglett.1c01775).
Another long-standing goal of the group, the simultaneous introduction of an alkyne and an azide onto a double bond, was realized in 2023. ABZ was used as a source of azido radicals under photoredox conditions. A fast addition to styrenes followed by oxidation ot the carbocation and reaction with alkynylboronates gave the desired azidoalkynylation products (Chem. Sci. 2023, 14, 9452-9460. DOI: 10.1039/D3SC03309K).
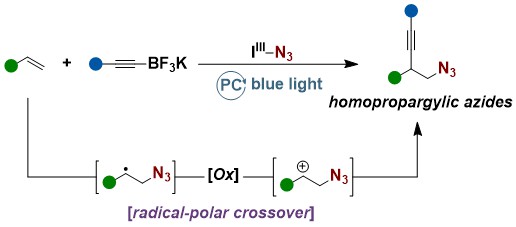
In 2024, we also described in details the background on how the reaction was discovered and optimized (Beilstein J. Org. Chem. 2024, 20, 701-713. 10.3762/bjoc.20.64).
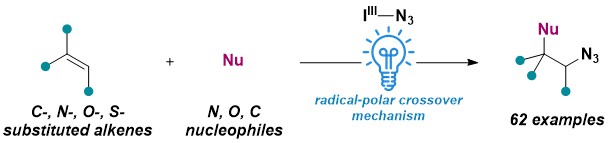
We then wondered if the developed radical-polar crossover azidation approach could be extended to further transformations. In fact, we developed in 2025 a general azidofunctionalization of styrenes, enamides and enol ethers enabling the addition of diverse O and N nucleophiles. Using only easily accessible alkenes and nucleophiles, we estimated that more than 1 million compounds could be accessed by this method. To enhance the chemical diversity of the obtained compounds, we used an unbiased clustering technique to select part of our scope entries. (Angew. Chem., Int. Ed. 2025, 64, e202420455. 10.1002/anie.202420455).
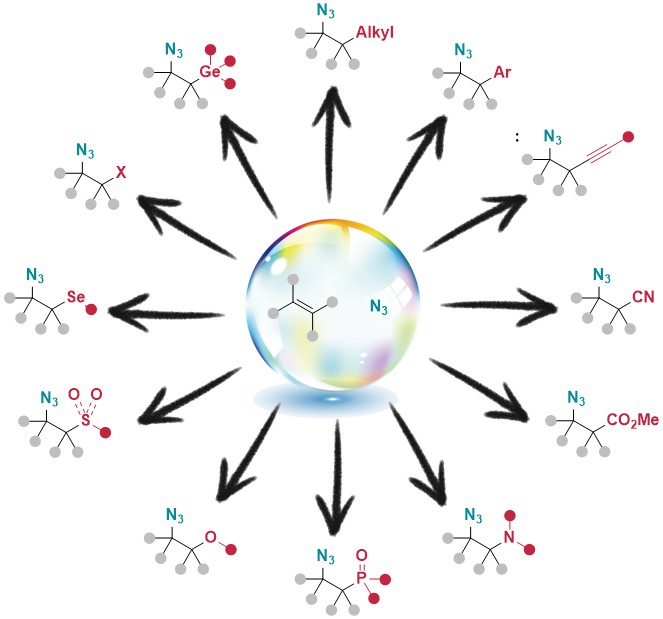
In general, azide chemistry is growing rapidly in organic synthesis and beyond, and many research groups have realized that hypervalent iodine reagents can be highly useful for azidation reactions. This motivated us to review this field in 2022 (Synthesis 2022, in press. DOI:10.1055/a-1966-4974).
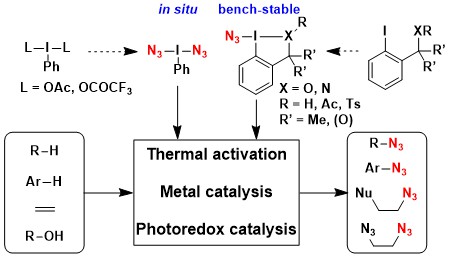
In particular, an impressive number of methods allowing the azidofunctionalization of alkenes with a broad diversity of functional groups have appaered since 2020. This motivated us to review this field in 2025 (ChemistryEurope 2025, e2500140. DOI: 10.1002/ceur.202500140).
Reactions with Cyanobenziodoxolone (CBX)
With cyanobenziodoxoles, we were able to develop a new method for the synthesis of thiocyanates starting from thiols (Chem. Eur. J. 2015, 21, 2662. DOI:10.1002/chem.201406171 ). As was the case for the related alkynylation reaction, this transformation is extremely efficient, with often quantitative yields obtained in a few minutes. Both the dimethyl and the carboxy reagents worked well. Many functional groups such as acids, alcohols or aromatic amines are tolerated. The cyanation of thiols displayed a broad generality, as it could be applied in the case of aromatic, heteroaromatic, aliphatic or glycosidic thiols.

After having developed the efficient alkynylation of carboxylic acids using EBX reagents under photoredox conditions, we wondered if a similar approach could be also successful for other benziodoxolones. In particular, the conversion of carboxylic acid is usually a multi-step procedure generating large amounts of waste. In 2016, we developed a one-step decarboxylative cyanation of carboxylic acid using CBX and an iridium photocatalyst. The reaction worked well for amino- and oxy- acids, but not for simple aliphatic acids. Mechanism investigations combining experiments and computational chemistry shown that the alkynylation and cyanation reaction, although similar on the first look, follow different mechanisms: The alkynylation proceeds via a radical, whereas the cyanation most probably involves an iminium intermediate (Chem. Sci. 2017, 8, 1790-1800. DOI:10.1039/C6SC04907A ).
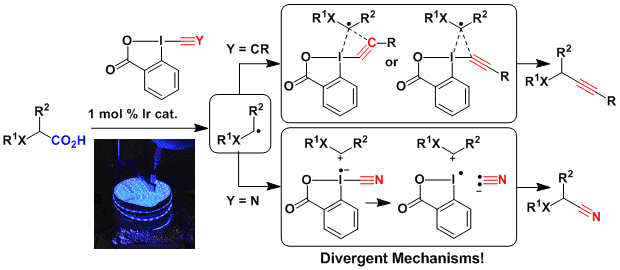
Interestingly, when protected amino acids were treated with CBX in absence of light, a different reaction occurred, resulting in a highly efficient synthesis of hydantoins. (Org. Lett. 2019, 21, 524-528. DOI:10.1021/acs.orglett.8b03843 )
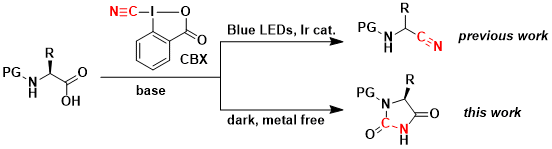
Enantioselective synthesis of tertiary azides and nitriles
The efficient azidation and cyanation of ketoesters derived from indanones with benziodoxol(on)e reagents gave access to the starting materials needed for an enantioselective decarboxylative allylation reaction. The corresponding homoallylic azides and nitriles could be obtained in high yield and enantioselectivity using a palladium catalyst and Trost’s chiral diphosphine ligands (Org. Lett. 2015, 17, 5832. DOI:10.1021/acs.orglett.5b03002 ).
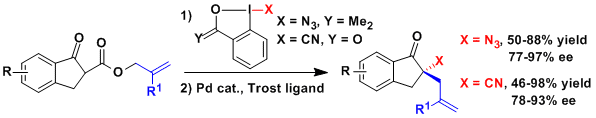
Umpolung of indoles and pyrroles
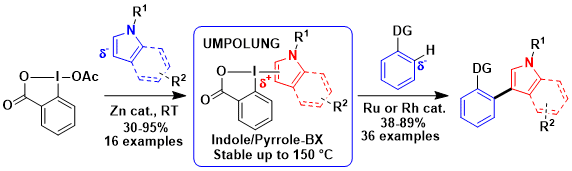
In 2017, an important breakthrough was achieved at LCSO with the discovery of Indole- and Pyrrole- BX reagents, the first examples of benziodoxoles with this type of heterocycles (Chem. Eur. J. 2017, 23, 14702. DOI:10.1002/chem.201703723 ). These new reagents were bench stable and could be used in the rhodium and ruthenium catalyzed C-H functionalization of arenes. These transformations could not be realized with other reagents. As often in science, the new reagents were discovered serendipitously when attempting new domino reactions (Helv. Chim. Acta 2017, 100, e1700221. DOI:10.1002/hlca.201700221 ). Later, the approach could be extended to the indolation of heterocycles, such as pyridones. (Beilstein J. Org. Chem. 2018, 14, 1208-1214. DOI:10.3762/bjoc.14.102 )
The approach was not limited to the C-H functionalization of (hetero)aromatic C-H bonds. In 2018, we showed that the directed functionalization of the C-H bond on the carbonyl group of benzaldehyde derivatives was also possible, using either an iridium or a rhodium catalyst for oxygen and nitrogen directing groups respectively. (Org. Lett. 2018, 20, 1473-1476. DOI:10.1021/acs.orglett.8b00337)
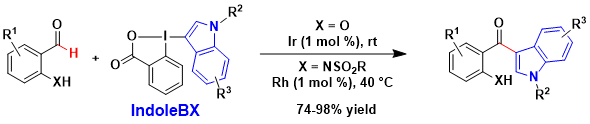
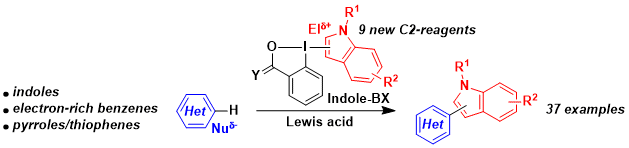
In 2018, a different mode of activation was discovered using silicon-based Lewis acids. These conditions allowed the coupling of the Indole- and Pyrrole-BX reagents with electron-rich (hetero)arenes with high regioselectivity. The method was particularly useful to access non-symmetrical bi-indole derivatives, which cannot be synthesized using existing oxidative coupling methods. During this work, a new procedure to access C2-substituted Indole-BX reagents was also developed. Chem. Eur. J. 2018, 24, 10049-10053. (DOI:10.1002/chem.201802142)
;”>
Vinyl Benziodoxolone Reagents
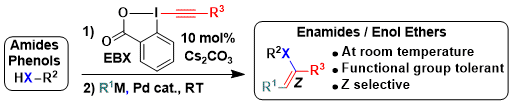
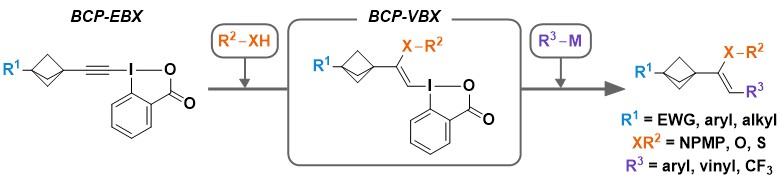
In comparison to EBX reagents, vinylbenziodoxolone (VBX) reagents have been less investigated. This was partly due to a lack of efficient synthesis, with the exception of the protocol reported by Olofsson and co-workers. In 2019, we reported a new approach for the synthesis of heteroatom-substituted VBX reagents by the base-catalyzed addition of phenols or sulfonyl amines onto EBX reagents. The catalyst loading was essential for an efficient synthesis and preventing follow-up reactions, as calculations showed that protonation of the obtained primary adduct was reversible and the intermediate could decompose or form the alkyne products. The reaction occurred under mild conditions, allowing the functionalization of drugs and amino acid derivatives. The high reactivity of the hypervalent iodine bond could be exploited in a stereoselective synthesis of enamides via palladium-catalyzed cross coupling reactions (Chem. Sci. 2019, 10, 3223-3230. DOI:10.1039/C8SC05573D ).
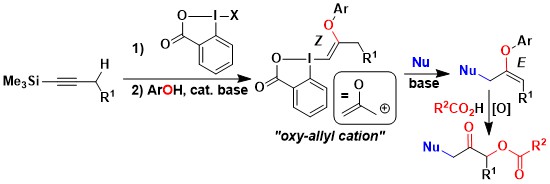
The obtained O-VBX reagents displayed very unusual reactivity under basic conditions in presence of nucleophiles: Substitution of the iodine was observed with simultaneous shift of the double bond. Such a reactivity is usually observed for oxy-allyl cation intermediates. Enol ethers were obtained with complete stereoselectivity, and their oxidation led to polyoxygenated compounds in few steps only starting from alkynes. (Angew. Chem., Int. Ed. 2020, 59, 18256-18260. DOI:10.1002/anie.202006707 ).
At this point, we were also interested to investigate the reactivity of VBX reagents in other transformations developed for the corresponding EBXs. In 2020, we demonstrated that VBX reagents also worked very well in the copper-catalyzed reaction with diazo compounds, resulting in an efficient oxyvinylation process. In this work, we also developed a new protocol for the efficient synthesis of alkyl-substituted VBX reagents. (Org. Lett. 2020, 22, 3884-3889. DOI:10.1021/acs.orglett.0c01150).
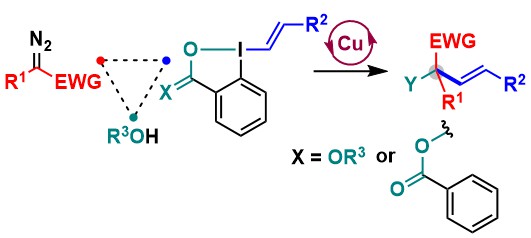
In 2023, we discovered an efficient trifluoromethylation of VBX reagents using high valent copper complexes. To promote the reaction, two conditions were developed: thermal activation at 120 °C or light irradiation. The reaction was successful both in batch and flow processes (Angew. Chem., Int. Ed. 2023, 62, e202306128. DOI: 10.1002/anie.202306128.
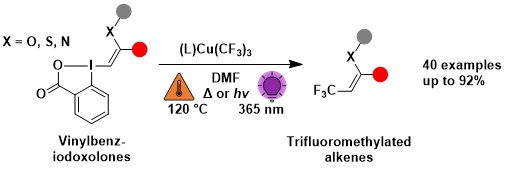
In 2025, we wondered if hypervalent iodine reagents could be used to further increase the chemical space of bicyclo[1.1.1] pentanes, which are currently of high interest in medicinal chemistry as bioisosteres of benzenes. We designed a new type of BCP-EBX reagents, which reacted efficiently with diverse types of N, O and S nucleophiles. The obtained VBX reagents could be then easily difversified using the methods previously developed in the group. (Angew. Chem., Int. Ed. 2025, 64, e202508404. 10.1002/anie.202508404.
Whereas VBX reagents were nearly unknown in 2015, the work of the groups of Olofsson, Nachtsheim, Yoshikai and our own group established them as usual reagents in the years 2016-2020. This motivated us to write a review in 2020 about this emerging class of reagents (Helv. Chim. Acta 2020, 103, e2000191 DOI:10.1002/hlca.202000191 ).
CpBx Reagents
A further milestone in the discovery of new reagents was achieved in 2024 with CyclopropenylBenziodoxoles. These reagents are easily accessed by simple addition of lithiated cyclopropenes onto iodine(III) precursors. They are especially excellent partners for gold catalysts in redox catalytic cycles, where they act as sigma-cyclopropenium synthetic equivalents. Under mild conditions, cyclopropene-including dienes and enynes could be obtained in close to quantitative yields. (Nat. Chem. 2024, 16, 901-912. 10.1038/s41557-024-01535-8).

These new CpBx reagents then enable the first efficient synthesis of 1,1′-bicyclopropenyls, one of the most neglected constitutional isomers of benzene. Synergistic gold/silver catalysis was used to activate both the hypervalent iodine bond of CpBx and the C-H bond of cyclopropene. (J. Am. Chem. Soc. 2024, 146, 29712-29719 10.1021/jacs.4c10996).

As a next application for CpBx reagents, we wondered if the copper-mediated trifluoromethylation methods developed for VBXs would be also successful in this case. Although thermal conditions could not be used, due to the lower stability of the reagents, the photo-mediated process was successful, leading to unique trifluoromethylated cyclopropene building blocks. The obtained products could be hydrogenated stereoselectively to the corresponding trifluoromethylated cyclopropanes, or converted to other cyclic and non-cyclic fluorinated building blocks. (ChemRxiv 2025. 10.26434/chemrxiv-2025-zpkg).

The fast expanding use of benziodoxole reagents
Togni’s work on trifluoromethylation and our work on alkynylation had amply demonstrated the utility of benziodoxole reagents. Most certainly, these reagents will be also highly useful for the transfer of other functional groups. Indeed, exciting new examples have started to emerge in azidation, cyanation, fluorination and many other transformations (Angew. Chem., Int. Ed. 2016, 55, 4436. DOI: 10.1002/anie.201509073 ).