Bacteria are micrometer-scale living organisms responsible for deadly diseases such as tuberculosis (still infecting 20% of the world population), but also producing a large amount of the oxygen we breathe. However, bacteria are so small that conventional light microscopes do not have a resolution high enough to image the cell sub-structures. We built a custom instrument to image bacteria in 3D with a nanometer resolution, using atomic force microscopy (AFM).
In collaboration with the laboratory of Prof. John McKinney, we are studying the growth and division of Mycobacteria, a genus of Actinobacteria including M. smegmatis and M. tuberculosis.
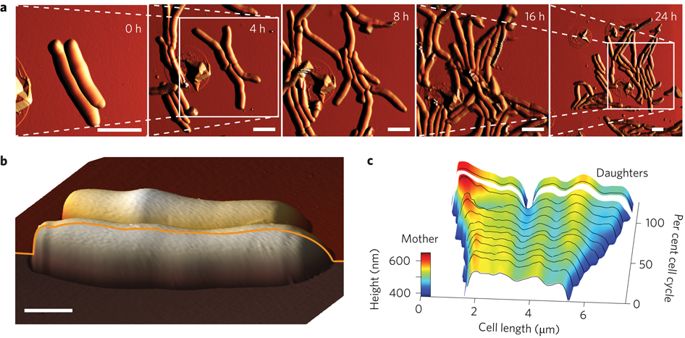
The way mycobacteria choose where to divide was previously unknown. AFM time-lapse imaging revealed nanoscale variations of the cell diameter along its length, called wave-troughs. The wave-troughs determine the future division site of the bacterium, up to three generations in advance. This raises fascinating new questions: how are the wave-troughs generated by the cell? how does the molecular machinery inside the cell recognize the wave-troughs to initiate division?
The mechanical stress in the bacterial cell wall can be measured with AFM, as cell wall under more tension will appear stiffer. During the process leading to division, a local increase of mechanical stress is visible in the bacterial cell wall, ultimately exceeding the ultimate strength of the cell wall and leading to cell separation is a few 10s of millisecond.

Growth symmetry or asymmetry of mycobacteria remains a controversial topic as opposing descriptions have been reported in the literature. Surprisingly, the AFM high-resolution time-lapses did not match any of the two previously reported models of pure symmetry and pure asymmetry. Mycobacterial pole growth dynamics was rather an intermediate between the two, reminiscent of the well-known “new end take off” mechanism for fission yeast. Using AFM nanomanipulation we show that growth asymmetry is not a physical occlusion phenomenon, but is inherent to mycobacterial growth.
Please note that the publication lists from Infoscience integrated into the EPFL website, lab or people pages are frozen following the launch of the new version of platform. The owners of these pages are invited to recreate their publication list from Infoscience. For any assistance, please consult the Infoscience help or contact support.
A biphasic growth model for cell pole elongation in mycobacteria
Nature Communications. 2020-01-23. Vol. 11, p. 452. DOI : 10.1038/s41467-019-14088-z.Please note that the publication lists from Infoscience integrated into the EPFL website, lab or people pages are frozen following the launch of the new version of platform. The owners of these pages are invited to recreate their publication list from Infoscience. For any assistance, please consult the Infoscience help or contact support.
Division site selection linked to inherited cell surface wave troughs in mycobacteria
Nature Microbiology. 2017. Vol. 2, p. 17094. DOI : 10.1038/nmicrobiol.2017.94.Please note that the publication lists from Infoscience integrated into the EPFL website, lab or people pages are frozen following the launch of the new version of platform. The owners of these pages are invited to recreate their publication list from Infoscience. For any assistance, please consult the Infoscience help or contact support.