Introduction
Benziodoxolones (BX), benziodoxoles (Bx) and benziodazolones (BZ) are high energy compounds. Therefore having good information on their stability is important for safety reasons. They are generally more stable than the corresponding acyclic reagents, but they should nevertheless be manipulated with great care. The goal of this website is to have all stability data on a single place instead of scattered in our different publications. We will be happy to add data collected by other laboratories.
Since 15 years we are working with these compounds, we had an accident and explosion only with azidobenziodoxolone (ABX, Zhdankin reagent, see accident report below). This compound has therefore to be manipulated with outmost care. Original DSC data are also available below. We thank Dr. Régis Mondière, Jerome Cassayre, Fides Benfatti, Ms. Marylene Stempien and Mr. Tony O’Sullivan from Syngenta Crop Protection Munchwilen AG for performing these measurements. We thanks Dr. Annik Nanchen, Mr. Christian Aeby, Ms. Delphine Berset and Mr. Daniel Steiner from TÜV SÜD Schweiz AG for performing the in-depth studies on azidation reagents, and DR. MER Thierry Meyer from the group of chemical and physical safety at EPFL for his expert analysis of the data. In 2024, thanks to the work of Mario Waser , Roland Obermüller and co-workers published in Org. Proc. Res. Dev. , we were pleased to be able to significantly extend the database.
For other benziodoxole reagents, stability data are not yet available: all these compounds should be therefore manipulated with great care.
According to their available stability data, we categorize the compounds in our laboratory as following:
Green: No compound, as all hypervalent iodine reagents are high energy compounds.
Yellow: Decomposition starts above 125 °C, and DSC peak height <3 W/g and peak width > 5 °C and energy of exotherm < 1000 J/g and no shock sensitivity and no explosion reported.
Orange: Decomposition starts between 90 and 125 °C or DSC peak height is 3-15 W/g or DSC peak width is 1-5 °C, or energy of exotherm between 1000 and 1500 J/g, and no shock sensitivity, and no explosion reported.
Red: Decomposition starts below 90 °C, or DSC peak height > 15 W/g, or DSC peak width < 1 °C, or shock sensitive, or energy of exotherm > 1500 J/g, or explosion reported.
Warning sign: Accident has occurred.
Please see the Waser/Obermüller work for a classification according to OREOS+.
!!The content of this site is purely informative. In no case it can replace risk/safety analysis in other laboratories by a safety specialist. The presented data are preliminary and do not constitute a full safety analysis. They therefore cannot replace in-depth safety analysis by specialized laboratories.!!
CpBx-reagents
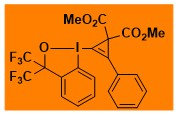 |
CpBX Decomposition starts: 75 °C DSC data: 1. peak height: 0.72 W/g, peak width: 72 °C, peak max: 122 °C, energy: 195 J/g, 2. peak height: 0.2 W/g, peak width: 24 °C, peak max: 207 °C, energy: 73 J/g, 3. peak height: 0.5 W/g, peak width: 130 °C, peak max: 379 °C, energy: 393 J/g. No shock sensitivity observed No explosion in more than 500 experiments Comment: Low safety hazard with this compound due to stepwise decomposition over a large temperature range, but the low temperature onset for decomposition requires some care and storage in the fridge. – |
EBX(x)-reagents
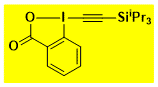 |
TIPS-EBX Decomposition starts: 135 °C DSC data: peak height: 2.3 W/g, peak width: 10.7 °C, peak max: 198 °C, energy: 497 J/g No shock sensitivity observed No explosion in more than 5000 experiments Comment: most often used EBX reagent, strong nucleophilic bases may lead to decomposition by silyl group removal – |
 |
TMS-EBX Decomposition starts: 117 °C DSC data: peak height: 3.2 W/g, peak width: 9.5 °C, peak max: 155 °C, energy: 596 J/g No shock sensitivity observed No explosion in more than 500 experiments Comment: sensitive to nucleophilic bases, as the silyl group is very easy to remove. Some batches decomposed in presence of bases, but never with explosive behavior – |
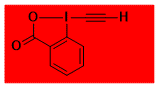 |
EBX Decomposition starts: -20 °C DSC data: not determined No explosion in more than 200 experiments Comment: unstable compound, needs to be generated in situ.Note: Itoh and co-workers later found that the compound is stable if co-crystallized with acetonitrile (Org. Lett. 2019, 21, 1098). – |
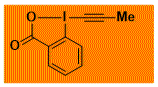 |
Me-EBX Decomposition starts: 97 °C DSC data: peak height: 4.0 W/g, peak width: 9.2 °C, peak max: 148 °C, energy: 697 J/g No shock sensitivity observed No explosion in more than 100 experiments – |
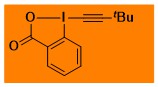 |
tBu-EBX Decomposition starts: 158 °C DSC data: peak height: 3.0 W/g, peak width: 10.6 °C, peak max: 195 °C, energy: 1066 J/g No shock sensitivity observed No explosion in more than 30 experiments – |
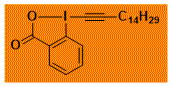 |
C14H29-EBX Decomposition starts: 115 °C DSC data: peak height: 1.7 W/g, peak width: 16 °C, peak max: 147 °C, energy: 485 J/g No shock sensitivity observed No explosion in more than 30 experiments – |
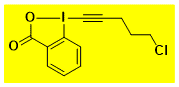 |
Cl(CH2)3-EBX Decomposition starts: 138 °C DSC data: peak height: 2.4 W/g, peak width: 16 °C, peak max: 151 °C, energy: 585 J/g No shock sensitivity observed No explosion in more than 30 experiments – |
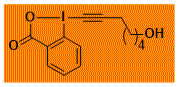 |
HO(CH2)5-EBX Decomposition starts: 124 °C DSC data: peak height: 1.7 W/g, peak width: 15 °C, peak max: 155 °C, energy: 438 J/g No shock sensitivity observed No explosion in more than 30 experiments – |
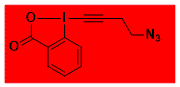 |
N3(CH2)2-EBX, JW-RF-010 Decomposition starts: 105 °C DSC data: peak height: 71 W/g, peak width: 1.2 °C, peak max: 129 °C, energy: 1927 J/g No shock sensitivity observed No explosion in more than 200 experiments – |
 |
Ph-EBX Decomposition starts: 113 °C DSC data: peak height: 1.9 W/G, peak width: 17 °C, peak max: 149 °C, energy: 559 J/g No shock sensitivity observed No explosion in more than 300 experiments – |
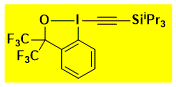 |
CF3-TIPS-EBx Decomposition starts: 140 °C DSC data: peak height: 0.87 W/g, peak width: 50 °C, peak max: 226 °C, energy: 342 J/g No shock sensitivity observed No explosion in more than 500 experiments |
Other benziodoxol(on)e reagents with an I-C bond
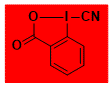 |
CBX Decomposition starts: 135 °C DSC data: peak height: 23 W/g, peak width: 2 °C, peak max: 152 °C, energy: 571 J/g No shock sensitivity observed No explosion in more than 500 experiments – |
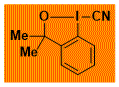 |
CBx Decomposition starts: 120 °C DSC data: peak height: 4.5 W/g, peak width: 10 °C, peak max: 133 °C, energy: 543 J/g No shock sensitivity observed No explosion in more than 100 experiments – |
 |
F3C-BX, Togni reagent II Data from the Waser/Obermüller database Decomposition starts: 110 °C DSC data: peak height: 7.8 W/g, peak width: 4.8 °C, peak max: 130 °C, energy: 574 J/g – |
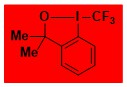 |
F3C-Bx, Togni Reagent I Data from the Waser/Obermüller database Decomposition starts: 78 °C DSC data: peak height: 0.25 W/g, peak width: 8.3 °C, peak max: 148 °C, energy: 740 J/g – |
 |
Ph-BX Data from the Waser/Obermüller database Decomposition starts: 167 °C DSC data: peak height: 1.4 W/g, peak width: 27 °C, peak max: 204 °C, energy: 464 J/g – |
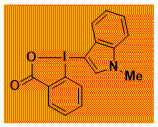 |
NMe-C3-IndoleBX Decomposition starts: 150 °C DSC data: peak height: 3.2 W/g, peak width: 3.7 °C, peak max: 183 °C, energy: 282 J/g No shock sensitivity observed No explosion in more than 100 experiments – |
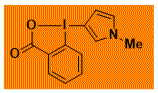 |
NMe-C3-PyrroleBX Decomposition starts: 190 °C DSC data: peak height: 11 W/g, peak width: 1.8 °C, peak max: 215 °C, energy: 574 J/g No shock sensitivity observed No explosion in more than 20 experiments – |
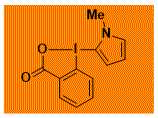 |
NMe-C2-PyrroleBX Decomposition starts: 165 °C DSC data: peak height: 4.9 W/g, peak width: 3.3 °C, peak max: 203 °C, energy: 546 J/g No shock sensitivity observed No explosion in more than 20 experiments |
Benziodoxol(on)es with an iodine-heteroatom bond
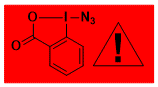 |
ABX, Zhdankin reagent Decomposition starts: 90 °C DSC data1 (pdf), DSC data2 (excel) : peak height: 78 W/g, peak width: 0.9 °C, peak max: 127 °C, energy: 1770 J/g Shock Sensitive. Confirmed by Fallhammer test, > 1 J (exact value too small to be determined) Friction Sensitive: positive for a friction load of 360 N in Alox plate Spark ignition successful (see video) Explosion! About 3 in 700 experiments, one resulting in injury. Accident Report. Comment: Highly useful and popular compound for azidation. Due to high hazard, should be manipulated with extreme care and in small quantities. – |
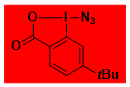 |
tBu-ABX Decomposition starts: 98 °C DSC data: peak height: 74 W/g, peak width: 0.7 °C, peak max: 139 °C, energy: 1440 J/g Shock Sensitive. Confirmed by Fallhammer test, > 2 J Friction Sensitive: negative for a friction load of 360 N in Alox plate No explosion in about 50 experiments. Comment: Slightly more stable derivative of Zhdankin reagent with similar reactivity and higher solubility in organic solvents. – |
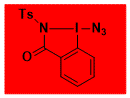 |
ABZ Decomposition starts: 143 °C, 268 °C DSC data: peak height: 25 and 1 W/g, peak width: 1.2 and 37.7 °C, peak max: 159 and 308 °C, energy: 965 and 380 J/g Shock Sensitive. Confirmed by Fallhammer test, > 5 J Friction Sensitive: negative for a friction load of 360 N in Alox plate No explosion in about 100 experiments. Comment: Most stable analogue of Zhdankin reagent, with only few residual shock sensitivity. Comparable reactivity in photoredox and metal catalysis, but thermal initiation requires higher temperature. Solubility limited in some organic solvents. For a detailed discussion, see: ChemRxiv – |
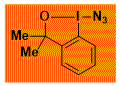 |
ADBX Decomposition starts: 107 °C DSC data: peak height: 2.7 W/g, peak width: 19 °C, peak max: 134 °C, energy: 843 J/g No shock sensitivity. Confirmed by hammer test. No explosion in more than 500 experiments Comment: Surprising strong effect on safety profile of replacing carboxy group. Should be used whenever possible for azidation. – |
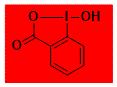 |
HO-BX Decomposition starts: 205 °C DSC data: peak height: 25 W/g, peak width: 0.5 °C, peak max: 229 °C, energy: 597 J/g No shock sensitivity observed No explosion in more than 500 experiments Comment: Detailed safety studies reported in Org. Proc. Res. Dev. 2013, 17, 318. Large temperature safety margin but still high explosive potential. – |
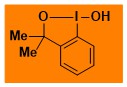 |
HO-Bx Data from the Waser/Obermüller database Decomposition starts: 101 °C DSC data: peak height: 3.1 W/g, peak width: 18 °C, peak max: 161 °C, energy: 683 J/g – |
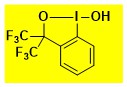 |
HO-CF3Bx Data from the Waser/Obermüller database Decomposition starts: 179 °C DSC data: peak height: 1.3 W/g, peak width: 19 °C, peak max: 240 °C, energy: 346 J/g – |
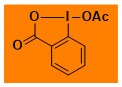 |
AcOBX Data from the Waser/Obermüller database Decomposition starts: 127 °C DSC data: peak height: 2.1 W/g, peak width: 17 °C, peak max: 211 °C, energy: 1445 J/g – |
 |
AcOBx Data from the Waser/Obermüller database Decomposition starts: 179 °C DSC data: peak height: 1.3 W/g, peak width: 19 °C, peak max: 240 °C, energy: 346 J/g – |
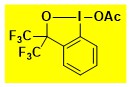 |
AcO-CF3Bx Data from the Waser/Obermüller database Decomposition starts: 148 °C DSC data: peak height: 0.70 W/g, peak width: 28 °C, peak max: 228 °C, energy: 315 J/g – |
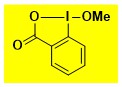 |
MeO-BX Data from the Waser/Obermüller database Decomposition starts: 145 °C DSC data: peak height: 1.8 W/g, peak width: 21 °C, peak max: 178 °C, energy: 486 J/g – |
 |
TfO-BX Data from the Waser/Obermüller database Decomposition starts: 164°C DSC data: peak height: 11.8 W/g, peak width: 2.1 °C, peak max: 188 °C, energy: 585 J/g – |
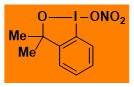 |
O2NO-Bx Data from the Waser/Obermüller database Decomposition starts: 116 °C DSC data: peak height: 7.2 W/g, peak width: 6.1 °C, peak max: 151 °C, energy: 731 J/g – |
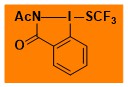 |
F3CS-BZ Data from the Waser/Obermüller database Decomposition starts: 108 °C DSC data: peak height: 0.1 W/g, peak width: 21 °C, peak max: 204 °C, energy: 93 J/g – |
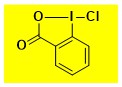 |
Cl-BX Data from the Waser/Obermüller database Decomposition starts: 137 °C DSC data: peak height: 2.2 W/g, peak width: 17 °C, peak max: 157 °C, energy: 490 J/g – |
 |
Cl-Bx Data from the Waser/Obermüller database Decomposition starts: 98 °C DSC data: peak height: 4.4 W/g, peak width: 3.8 °C, peak max: 112 °C, energy: 264 J/g – |
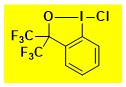 |
Cl-CF3Bx Data from the Waser/Obermüller database Decomposition starts: 189 °C DSC data: peak height: 0.92 W/g, peak width: 20 °C, peak max: 219 °C, energy: 250 J/g – |
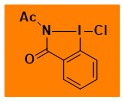 |
Cl-AcBZ Data from the Waser/Obermüller database Decomposition starts: 157 °C DSC data: peak height: 6.8 W/g, peak width: 1.1 °C, peak max: 171 °C, energy: 197 J/g – |
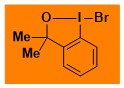 |
Br-Bx Data from the Waser/Obermüller database Decomposition starts: 77 °C DSC data: peak height: 1.4 W/g, peak width: 7.7 °C, peak max: 96 °C, energy: 143 J/g – |
 |
Br-CF3Bx Data from the Waser/Obermüller database Decomposition starts: 156 °C DSC data: peak height: 1.3 W/g, peak width: 5.4 °C, peak max: 204 °C, energy: 209 J/g – |
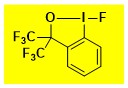 |
F-CF3Bx Data from the Waser/Obermüller database Decomposition starts: 171 °C DSC data: peak height: 0.81 W/g, peak width:29 °C, peak max: 225 °C, energy: 315 J/g – |