In the context of NCCR Catalysis, our group recently initiated collaborations with the Corminboeuf and the Laino groups to increasingly use digital tools in the organic chemistry laboratory. Our first publications in this area jointly with the Corminboeuf group appeared in 2024.
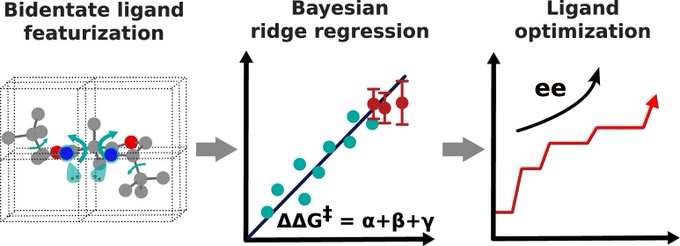
Motivated by our use of chiral bidentate ligands, such as bisoxazolines, a reaction-agnostic featurization of bidentate ligands was first developed. This approach enable a Bayesian ridge regression for enantioselectivity, enabling ligand optimization (ACS Catal. 2024, 14, 9302-9312 10.1021/acscatal.4c02452)
In a second work, we adapted Bayesian reaction optimization to take the cost of experiments into consideration. This way, the experimentalist will be suggested the lest costy experiments leading to improvement of the desired reaction parameters. Cost can be based on market prices, ease of synthesis or availability in laboratory (Digital Discovery 2024, 3, 2289-2297 10.1039/D4DD00225C).
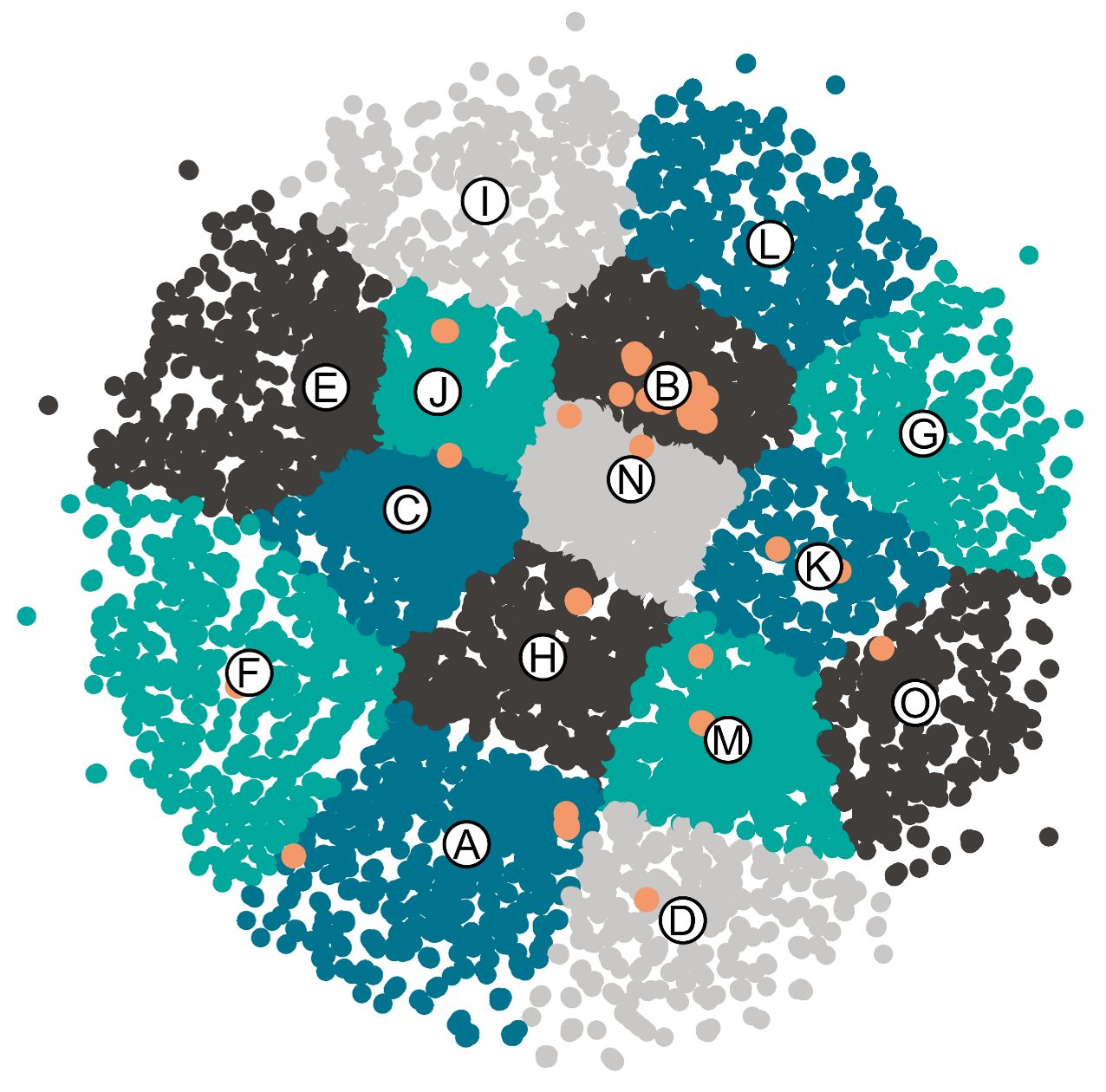
Together with experimental work, we are also interested to develop digital tools to select entries for scope in organic transformations. In the context of our azidofunctionalization of alkenes, using only easily accessible alkenes and nucleophiles, we estimated that more than 1 million compounds could be accessed. To enhance the chemical diversity of the obtained compounds, we used an unbiased clustering technique to select part of our scope entries. (Angew. Chem., Int. Ed. 2025, 64, e202420455. 10.1002/anie.202420455).
Reaction optimization is another field where digital tools become increasingly important. In our work on the Pd-catalyzed carboetherification of cyclopropenes, we used multivariate linear regression (MLR) models built by featurized monophosphine ligands from the Kraken data set to predict the yield of the reaction. The results indicated that no other ligand in the dataset was expecting to lead to better yields, allowing us to focus on other optimization factors (Chem. Sci. 2026, 17, in press. DOI: 10.1039/D5SC09351A ).