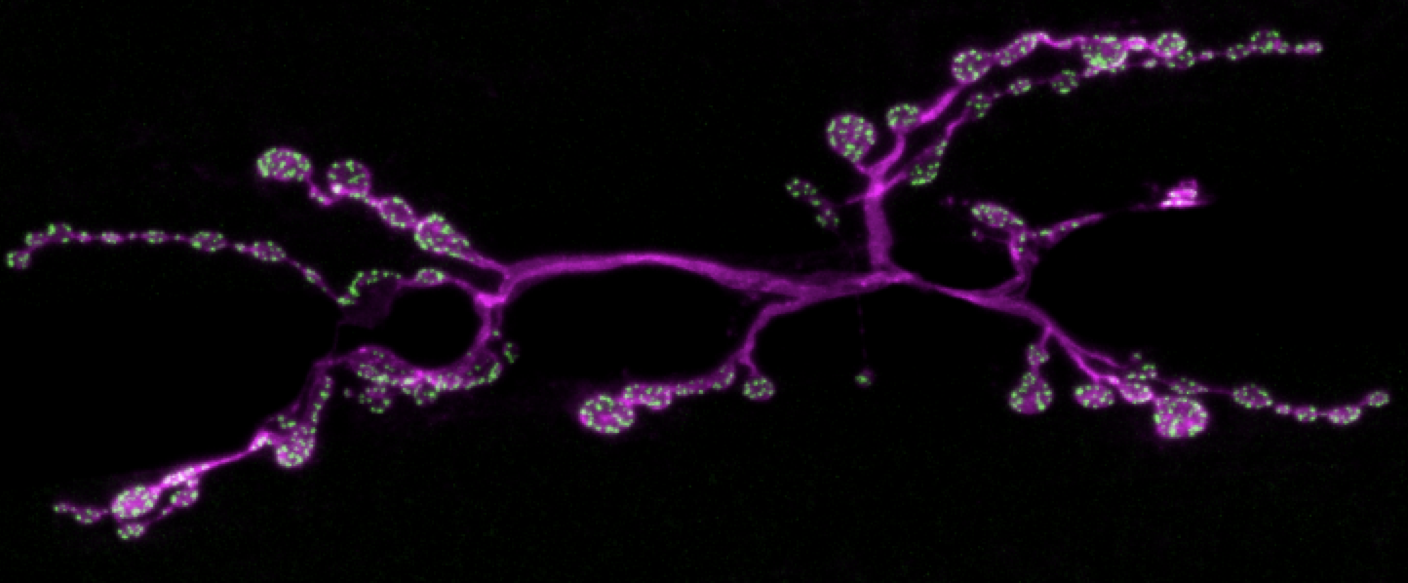
Synapses in Motor Circuits
Our laboratory investigates the biology and disorders of synapses – the specialized connections that allow communication between neurons within circuits – interconnected groups of neurons which through concerted action enable behaviour, most often by altering motor activity. Synapses are not static structures – but dynamically change in function, complexity and constituent numbers during development and throughout adult life. Many of the most remarkable capabilities of the nervous system are thought to be made possible by this plastic ability to change and react in response to experience.

Motor circuits receive and process input from sensory and central neurons and via motor neurons relay commands to muscles to produce behaviour. To execute these functions efficiently, many components of the motor system are exquisitely specialised. For example, neuromuscular junction (NMJ) synapses between motor neurons and muscles are adapted to amplify small neuronal impulses into large muscle contractions. The ancient and fundamental necessity of the motor system for complex animal life, the ability to image motor NMJ and other synapses in vivo, and the high accuracy of locomotor behavioural quantification, make motor circuits an ideal system to develop a molecular and cellular understanding of how synapses function, adapt and coordinate to produce ensemble activity.
Neurogenetic models

Visualisation of Drosophila brain, nerve cord and motor neurons
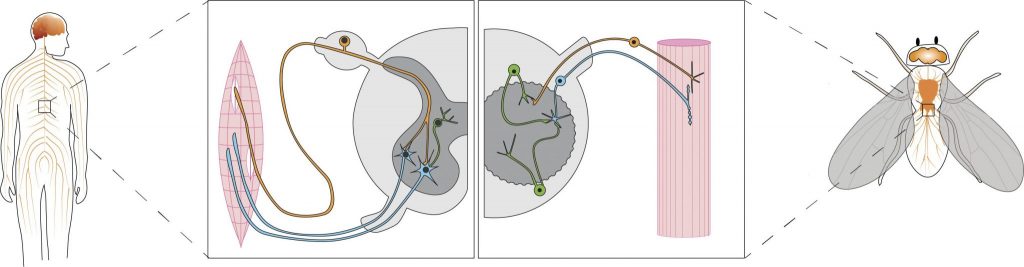
Mechanisms of Motor System Disease
Historically, invertebrate genetic models such as Drosophila have provided insight into human physiology and disease through basic research into fundamental biological mechanisms. However, in the post-genomic era, realisation of the extraordinary degree of genetic conservation between humans and invertebrates has prompted the development of Drosophila models of human disease in order to conscript the sophisticated genetic and cellular tools available in this system. We have further enhanced the fidelity of these models of human disease by using genome engineering technology. Our goal is to exploit these models to develop new insight into the molecular and cellular mechanisms of motor system disorders such as Spinal Muscular Atrophy (SMA), Amyotrophic Lateral Sclerosis (ALS), also known as motor neuron disease (MND), and Frontotemporal dementia (FTD) in order to develop novel therapeutic strategies, including through the use of gene therapy.
Current Research Topics
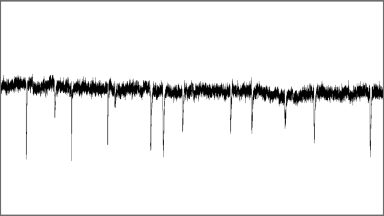
Miniature Neurotransmission
Miniature neurotransmission or ‘minis’ have been observed at every fast chemical synapse ever examined. Previously dismissed as ‘noise’, we have discovered that miniature neurotransmission is in fact a parallel trans-synaptic signal with unique regulation and critical functions.
Neurotransmission Circuit Logic
Neurons can employ an array of neurotransmitters to activate, inhibit or modulate interacting neurons within circuits. Using new neurogenetic tools, we are investigating the logic of how these neurotransmitters are deployed.
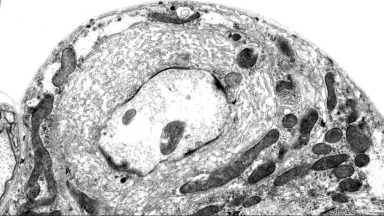
Activity-dependent plasticity
Synapses and circuits adapt to changes in activity in order to optimise behavioural output. Using a novel paradigm for activity-dependent plasticity, we are investigating the molecular regulation of this process.
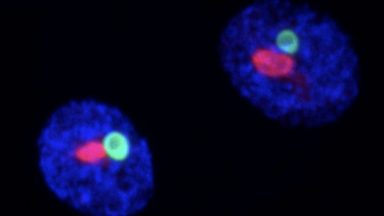
Mechanisms of ALS
A diverse range of proteins have been linked to genetic forms of the motor neuron disease ALS. Using genome engineering, we have generated novel ALS models in order to identify an underlying molecular genetic logic for this devastating disease.
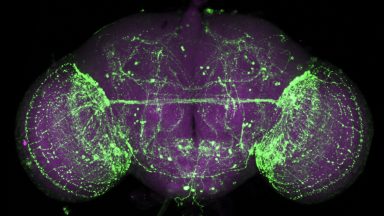
Ageing and Neurodegeneration
In neurodegenerative disorders synapses degenerate, axons retract and eventually neurons die disrupting brain circuits. Using new model systems, we are investigating the cell biology of this process during normal ageing and in models of human neurodegenerative disease.
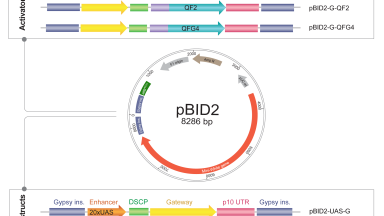
Neurogenetic tools
Neuroscience has benefited from increasingly sophisticated genetic tools and methods. For our studies, we design and build improved Drosophila genetic engineering tools and continue to develop improved methods to image and manipulate synapses and circuits.

Master Research Projects
For Master Project possibilities, go to our Opportunities page.