The “language” of endosymbiotic partners.
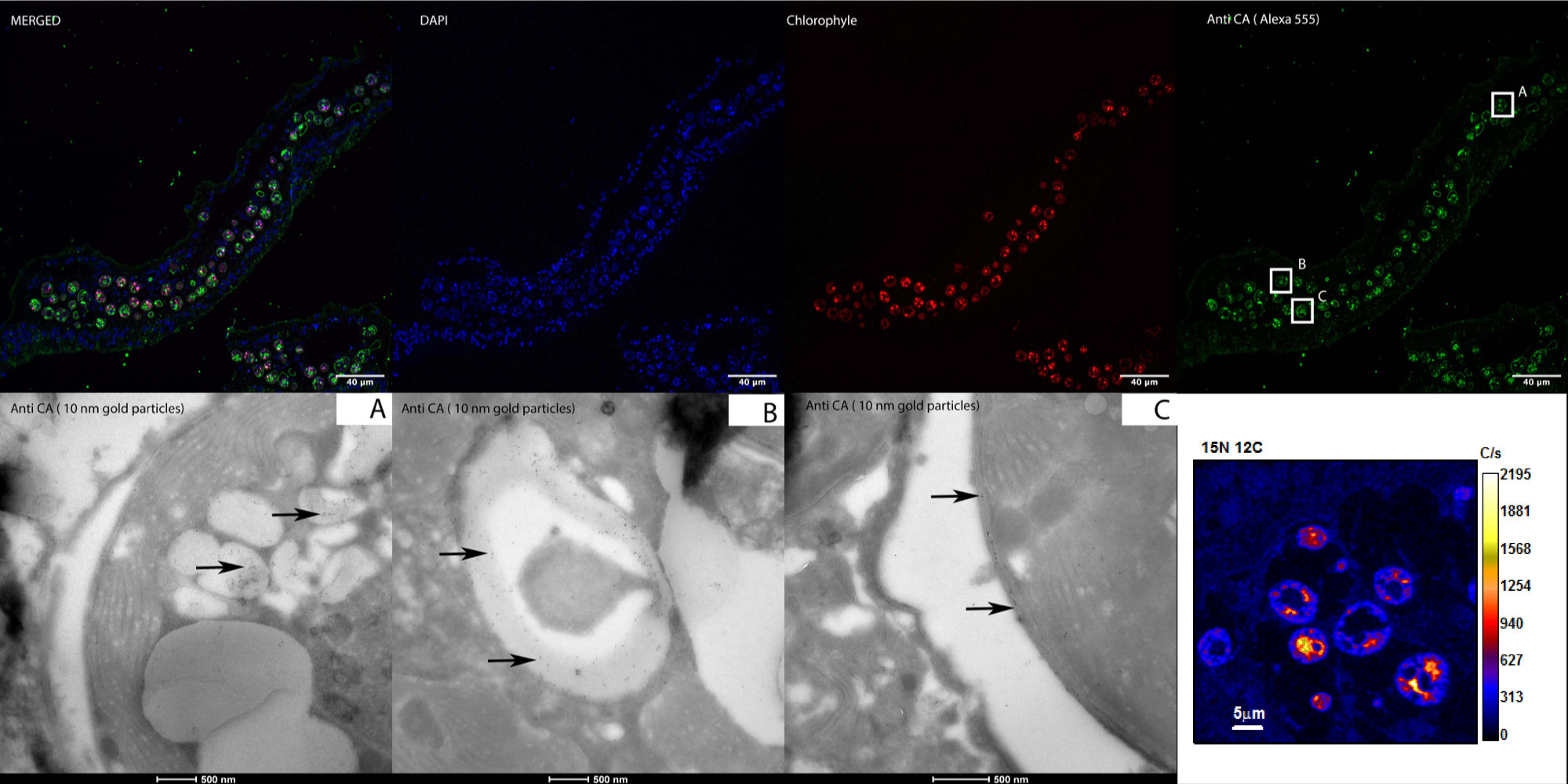
A correlative workflow combining cryo-preparation, fluorescence/electron microscopy, and NanoSIMS imaging to “listen” to the molecular and metabolic dialogue between partners in symbiotic corals.
Climate change threatens populations and ecosystems all over our planet. A direct consequence of the warming of the oceans is coral bleaching. Coral reefs support the most biologically diverse marine ecosystems and are extremely sensitive to changes to their physical environment (Baker et al., 2008). Mass bleaching events have been accelerating dramatically over the last decade, manifested through a loss of the endosymbiotic photosynthesizing algae hosted by the coral organisms. In homeostatic conditions, these pigmented dinoflagellates algae translocate photosynthates to their coral host, and thus provide an essential component of the corals’ nutritional requirements. This symbiosis thus keeps the corals alive in the typically highly oligotrophic (nutrient-poor) tropical ocean conditions (Kopp et al., 2015; Krueger et al., 2018). Prolonged bleaching leads to the death of the coral host, essentially from starvation (Hoegh-Guldberg, 1999), and has dramatic consequences for the full reef ecosystem that individual corals support. Entry and establishment of an endosymbiotic relationship involves a sequence of highly controlled stages that are linked to the exchange of nutrients and molecules that permit the symbionts to survive inside the host and modulate host metabolism.
In order to study the fundamental process of endosymbiosis and its metabolic impact, we have developed a correlative workflow that stretches from cryo-preparation (Loussert Fonta et al., 2015) to ion microprobe imaging; NanoSIMS in particular. By avoiding classical protocols including ethanol dehydration and resin embedding, this technique reduces loss of soluble cellular content and minimizes dilution of isotopic enrichments, in particular 13C, thereby enhancing the quality of NanoSIMS images.
We are obtaining new data on the symbiotic coral Stylophora pistillata and its symbiont algae Symbiodinium prepared by this cryo-based workflow by adding new information on protein localization. Our methodology has strong potential in the study of field of symbiotic organisms and in the field of biological imaging in general.
References:
Baker, A.C., Glynn, P.W., and Riegl, B. (2008). Climate change and coral reef bleaching: An ecological assessment of long-term impacts, recovery trends and future outlook. Estuar. Coast. Shelf Sci. 80, 435–471.
Hoegh-Guldberg, O. (1999). Climate change, coral bleaching and the future of the world’s coral reefs. Mar. Freshw. Res. 50, 839.
Kopp, C., Domart-Coulon, I., Escrig, S., Humbel, B.M., Hignette, M., and Meibom, A. (2015). Subcellular Investigation of Photosynthesis-Driven Carbon Assimilation in the Symbiotic Reef Coral Pocillopora damicornis. MBio 6.
Krueger, T., Bodin, J., Horwitz, N., Loussert-Fonta, C., Sakr, A., Escrig, S., Fine, M., and Meibom, A. (2018). Tissue- and cellular-level allocation of autotrophic and heterotrophic nutrients in the coral symbiosis — A NanoSIMS study.
Loussert Fonta, C., Leis, A., Mathisen, C., Bouvier, D.S., Blanchard, W., Volterra, A., Lich, B., and Humbel, B.M. (2015). Analysis of acute brain slices by electron microscopy: A correlative light–electron microscopy workflow based on Tokuyasu cryo-sectioning. J. Struct. Biol. 189, 53–61.
Tokuyasu, K.T. (1973). A technique for ultracryotomy of cell suspensions and tissues. J. Cell Biol. 57, 551–565.