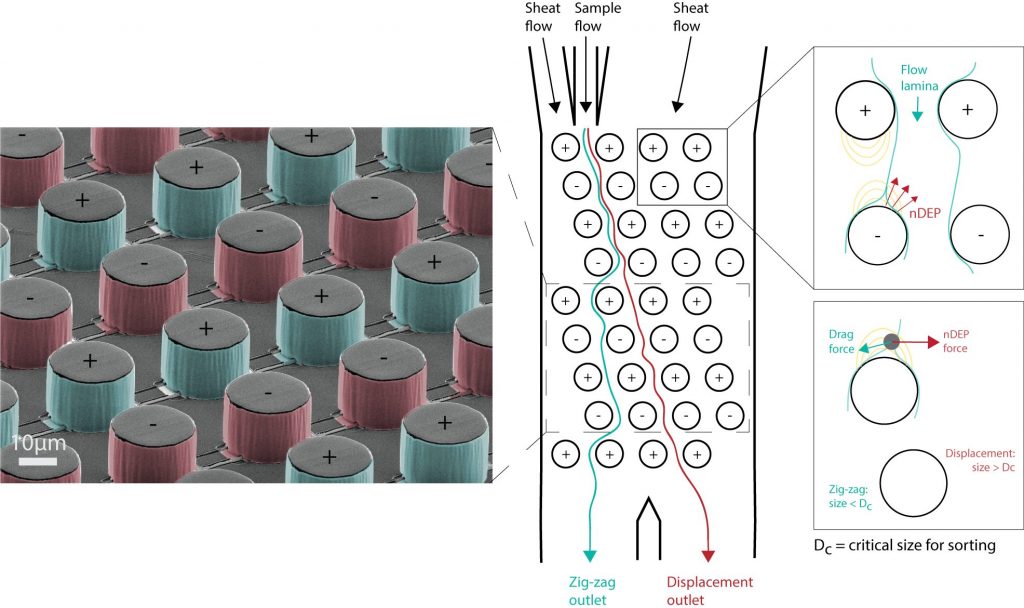
Deterministic Lateral Displacement (DLD) is a well-established microfluidic approach for sorting particles based on their mechanical properties. DLD is a purely passive technique based on regular arrangements of pillars. The in-flow particle trajectories are univocally determined by their size and by the geometry of the array. DLD is a very powerful tool for particle separation, several studies already demonstrated large sorting throughput and purity, mainly in the micrometre range, for example, to sort cells or bacteria. However, in order to sort smaller particles a nano-sized gap between the DLD pillars is needed, which requires challenging fabrication and makes the fluidics prone to clogging issues.
In the Guiducci lab, we recently introduced a microfabrication process to embed three-dimensional electrodes in microfluidics for dielectrophoretic (DEP) applications. These active pillars can be employed to replace the passive DLD posts obtaining a DEP-assisted DLD device, where the particles’ trajectories can be electrokinetically tuned in real-time. This method has been validated on polystyrene beads in low conductivity media, in collaboration with NanoLund, the Center for Nanoscience at Lund University, Sweden [1]. The sorting size of a passive DLD device has been reduced 25 times thanks to the application of the electric field. We are currently testing different electrodes shapes and configurations to sort particles down to nanometres in size.
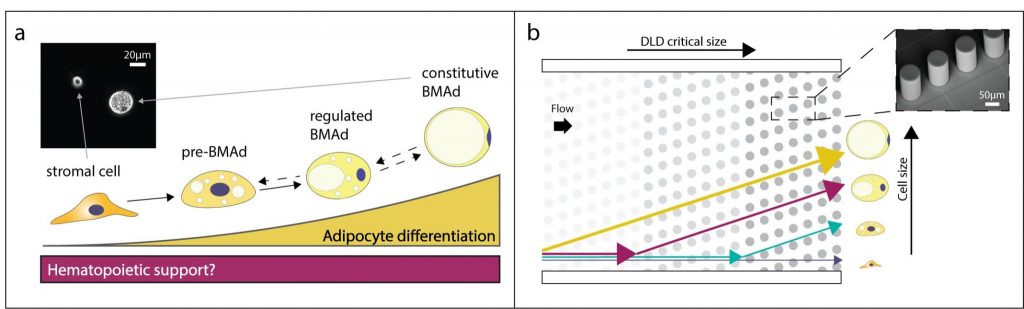
One main drawback of DLD is the lack of versatility, as the sorting output is fixed by the array geometry. The association of DLD with electrically-active pillars enables instead for a dynamic selection of output fractions. We propose this technology for the isolation of bone marrow adipocytes (BMAds) at different stages of differentiation based on their difference in size, as shown in Fig.2. In fact, despite being the most abundant stromal cell type within the bone marrow, the function of bone marrow adiposity and its effect on blood formation remain unclear. Contradictory results could be explained by the difficulty in isolating BMAds at specific stages of maturation. Our tool offers biologists the opportunity to characterize well-defined stages of differentiation, to better discern their function within the bone marrow niche and their impact on blood formation. This could ultimately pave the way to novel therapies for several pathologic conditions, such as leukemia or bone and metabolic diseases. This study is held in collaboration with the Laboratory of Regenerative Hematopoiesis at UNIL, Lausanne [2].