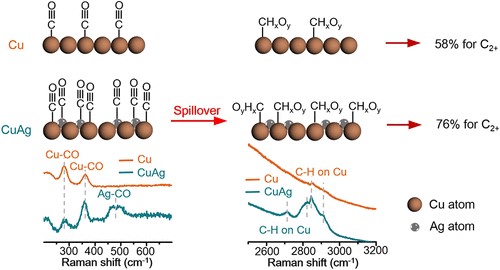
Solar energy is the ultimate energy source for our human beings. In nature, the solar flux is absorbed by plants and stored in chemical bonds. Mimicking nature, artificial photosynthesis is the process of converting abundant molecules such as CO2 and H2O to value-added chemicals using solar energy. This becomes the primary research focus of the solar fuel group in LPI. The major two approaches are photoelectrochemical conversion (PEC) and electrocatalysis powered by photovoltaics (PV-EC). The reactions that we investigated are water reduction to hydrogen and CO2 electroreduction to chemicals/fuels.
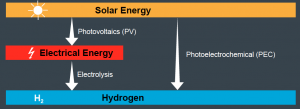
Building on our expertise of semiconductor science and device fabrication, we aimed at developing photoelectrodes based on earth abundant materials for these important chemical reactions. We are also interested in designing novel electrocatalysts and fabricating PV-EC devices for selective and stable CO2 reduction to CO, C2H4 and other liquid fuels. Operando Raman and operando synchrotron techniques are the state-of-the-art tools that we used for gaining mechanistic insights into the electrode/electrolyte interface. Recently, we plan to work on integrating electrolyzer with fermenter for producing C2-C4 alcohols from CO2 and H2O. Our work can be divided into three main topics:
- Fabricating photoelectrochemical devices, mainly Cu2O-based photoelectrodes, for water reduction and CO2 reduction
- Designing electrocatalysts for selective CO2 reduction to value-added chemicals and engineering PV-electrolyzer systems based on these novel catalysts
- Employing state-of-the-art operando techniques, including Raman, Synchrotron and Infrared, to investigate the catalytic interfaces under reaction conditions