
Predicting the fate of substances in the atmosphere requires understanding of its partitioning behavior among gas and various condensed phases (solid, semisolid, aqueous and organic liquids). Acidity of aerosols is a consequence of phase partitioning and chemical equilibrium among species present in the aqueous phase. We model relevant behavior at the molecular scale using discrete particle (molecular dynamics) simulations, or with semi-empirical parameterizations within the framework of classical chemical thermodynamics. A computationally-efficient model for inorganic thermodynamic calculations (ISORROPIA II) – the de facto module for gas/particle partitioning and acidity calculations used in atmospheric chemical transport models – is developed and supported by members at LAPI.
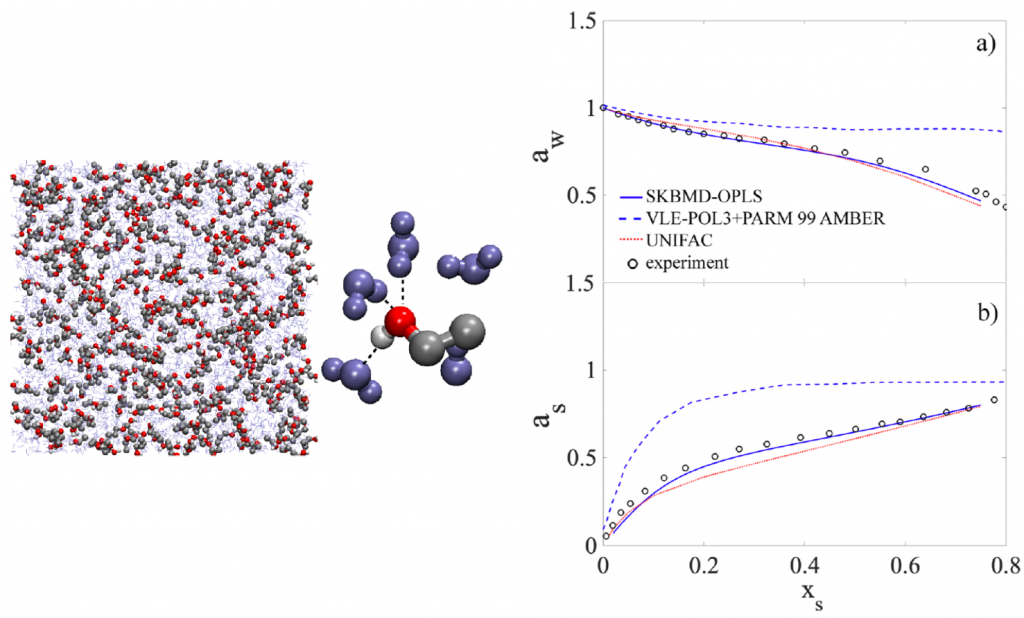
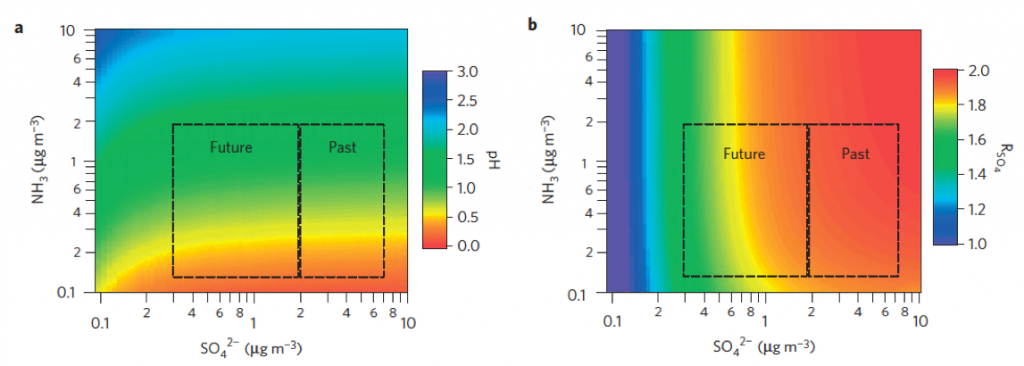
Representative publications.
- Lbadaoui-Darvas et al., J. Phys. Chem. B, doi:10.1021/acs.jpcb.9b06735, 2019.
- Vasilakos et al., Atmos. Chem. Phys., doi:10.5194/acp-18-12765-2018, 2018
- Guo et al., Atmos. Chem. Phys., doi:10.5194/acp-18-12241-2018, 2018.
- Ingall et al. Atmosphere., doi:10.3390/atmos9050201, 2018.
- Shi et al., Environ. Sci. Tech., doi:10.1021/acs.est.6b05736, 2017.
Software.
ISORROPIA and ISORROPIA II: models calculate the composition and phase state of an ammonia -sulfate -nitrate -chloride -sodium -calcium -potassium -magnesium -water inorganic aerosol in thermodynamic equilibrium with gas phase precursors.