J. Xu, Y. M. Liu, A. S. Lipton, J. Ye, G. L. Hoatson, P. J. Milner, T. M. McDonald, R. L. Siegelman, A. C. Forse, B. Smit, J. R. Long, and J. A. Reimer, Amine Dynamics in Diamine-Appended Mg2(dobpdc) Metal–Organic Frameworks J Phys Chem Lett (2019) doi: 10.1021/acs.jpclett.9b02883
Abstract
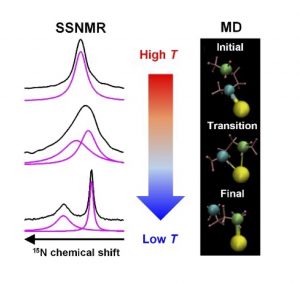
Variable-temperature 15N solid-state NMR spectroscopy is used to uncover the dynamics of three diamines appended to the metal–organic framework Mg2(dobpdc) (dobpdc4– = 4,4′-dioxidobiphenyl-3,3′-dicarboxylate), an important family of CO2 capture materials. The results imply both bound and free amine nitrogen environments exist when diamines are coordinated to the framework open Mg2+ sites. There are rapid exchanges between two nitrogen environments for all three diamines, the rates and energetics of which are quantified by 15N solid-state NMR data and corroborated by density functional theory calculations and molecular dynamics simulations. The activation energy for the exchange provides a measure of the metal−amine bond strength. The unexpected negative correlation between the metal−amine bond strength and CO2 adsorption step pressure unravel that metal-amine bond strength is not the only important factor in determining the CO2 adsorption properties of diamine-appended Mg2(dobpdc) MOFs.