 |
 |
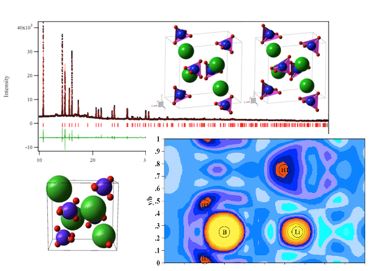 |
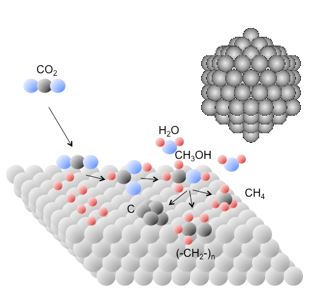
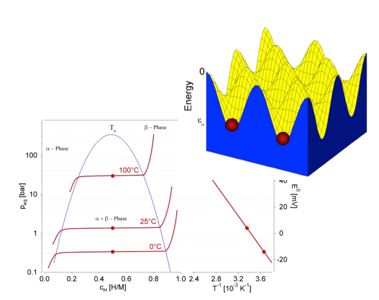
| Surface science, NAP-XPS Clusters, Reactions | Thermodynamics, TDS Lattice gas, Adsorption | Structure, electron density distribution |
 |
 |
 |
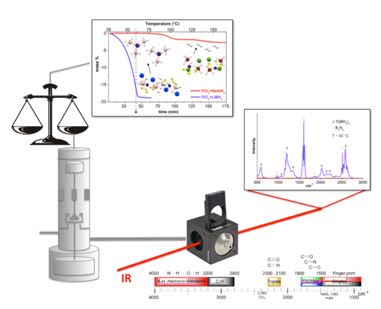
| In-situ Raman, TERS, and IR spectroscopy | Synthesis of intermetallic compounds, complex hydrides nano materials | Electrochemistry DoS, Electrolysis |
.
Few materials allow to store energy with a similar energy density as compared to the energy density of fossil fuels. Some hydrides, ammonia, and synthetic hydrocarbons reach an energy density of 10 kWh/kg. Batteries today allow storing 0.2 kWh/kg, however, there is a potential to increase the energy density of new advanced batteries to approx. 1 kWh/kg.
The past research activities have focused on the interaction of hydrogen with metals and intermetallic compound for the application in metal hydride batteries, hydrogen in Pd-Clusters and hydrogen adsorption on carbon nanostructures incl. carbon nanotubes. Ten years ago we started to intensively investigate complex borohydrides for hydrogen storage as the first group worldwide. Today borohydrides are the subject of intense research around the globe. Our activities are focused on the synthesis of the materials, the characterization by means of X-ray (synchrotron) and neutron diffraction, photoelectron-, infrared-, and Raman-spectroscopy. The dynamics of the hydrogen in the bulk and at the surface is investigated by gas ad- and absorption isotherms (mass flow) and micro-balance, nuclear magnetic resonance (NMR), and quasi-elastic neutron scattering, thermal desorption spectroscopy (mass spectroscopy), caloriemetry and isotope labeling (H, D). We plan to install a near ambient pressure X-ray photoelectron spectrometer (NAP-XPS) for the investigation of the surface composition and chemical state of materials interacting with hydrogen and CO2. Furthermore, Tip enhanced Raman Spectroscopy (TERS) will allow us to investigate single molecules at the surface of the catalyst.
- Schlapbach, L; Zuttel, A, ‘Hydrogen-storage materials for mobile applications’, Nature 414:6861 (2001), pp. 353 – 358
- Zuttel, A; Remhof, A; Borgschulte, A; Friedrichs, O, ‘Hydrogen: the future energy carrier’, Philosophical Transactions Of The Royal Society A-Mathematical Physical And Engineering Sciences 368:1923 (2010), pp. 3329 – 3342.
- Zuttel, A; Chartouni, D; Gross, K; Bachler, M; Schlapbach, L, ‘Structural- and hydriding-properties of the Zr(V0.25Ni0.75)(alpha) (1<=alpha<=4) alloy system’, Journal Of Alloys And Compounds 253: (1997), pp. 587 – 589.
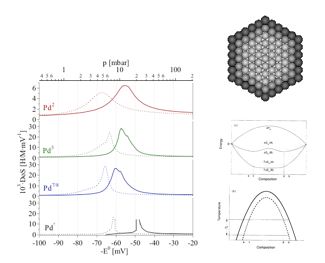
- Zuttel, A; Nutzenadel, C; Schmid, G; Chartouni, D; Schlapbach, L, ‘Pd-cluster size effects of the hydrogen sorption properties’, Journal Of Alloys And Compounds 293: (1999), pp. 472 – 475.
- Zuttel, A; Sudan, P; Mauron, P; Kiyobayashi, T; Emmenegger, C; Schlapbach, L, ‘Hydrogen storage in carbon nanostructures’, International Journal Of Hydrogen Energy 27:2 (2002), pp. 203 – 212.
- Zuttel, A; Wenger, P; Rentsch, S; Sudan, P; Mauron, P; Emmenegger, C, ‘LiBH4 a new hydrogen storage material’, Journal Of Power Sources 118:40210 (2003), pp. 1 – 7.