Lipid Droplets and Liposomes

#unfortunately nothing here
Nano- and micro-scale aqueous interfaces
Water. No substance on earth is so intimately linked to our well-being. Without it, we die. On a more scientific level, without water, membranes -the structures that provide the architecture of our cells and organelles- cannot function. Charges and charged groups cannot be dissolved, self-assembly cannot occur, and proteins cannot fold. Apart from the intimate link with life, water also shapes the earth and our climate. Our landscape is formed by slow eroding/dissolving processes of rocks in river and sea water; aerosols and rain drops provide a means of transport of water. Our society depends on products that all relate to water and aqueous systems, such as food products, medicine, and consumer goods.
In most of the above mentioned systems it is the interfacial region (of the membrane, the droplet, or the particle) that determines much of the physical, chemical, biological, and geological properties. Interfacial water is often considered in one of two ways: As a background, describable by a single parameter, or simply omitted. Alternatively, it is studied in great detail in an environment or condition that is precisely defined but so oversimplified that it has little to do with the real world. Aqueous interfaces are mostly studied in vacuo, or as a planar water/air interface.
However, interfacial water exists on different length scales, from sub-nanometer to micron sized (corrugations, organelles, membranes, liposomes), and is often buried inside another solid or liquid environment that is not at all comparable to vacuum or air. It can also be subject to conditions of flow or electrostatic fields. The absence of molecular knowledge of realistic interfaces is due to a lack of tools that can access buried nano- or microscopic interfaces in liquids and solids. Research at LBP is aimed to develop non-invasive label-free optical tools that can probe aqueous systems and interfaces on different length scales and to use these tools to understand the molecular level properties of water in diverse systems such as in solutions, at buried interfaces, in and outside droplets, and curved nanoscale membranes (illustrated in the below Figure).
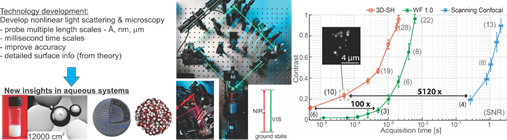
The LBP approach to study aqueous systems with a multiscale toolbox. Left: Rationale of our work, and illustrations of the studied systems. Middle: Photos of parts of the SFS systems (left inset), the first SHG wide field microscope (WF 1.0), and an optical scheme for SHS (right inset). For SFS the excitation is done with an infrared pulse at resonance with a vibrational mode and a non-resonant visible pulse. Right: Throughput curves for SH imaging of 50 nm BaTiO3 particles on a glass cover slip, measured with our homebuilt wide field systems “3D-SH’, ‘WF 1.0’ and a commercial device (‘scanning confocal’ Leica SP5, EPFL microscopy facility).
A variety of systems have been studied, comprised of most notably, emulsions (oil droplets in water and water droplets in oil) and membrane interfaces of liposomes. We find in most studies that the length scale of the system determines interfacial structure and that the chemical interfacial details drive the outcome of many processes.
Highlights:
ACS Nano, (2017) 111, 12111-12120
Nat. Commun., (2017), 8, 15548-1-6
Chem. Phys. Lett. Frontiers, (2014), 615, 124-130
Angew. Chem. Int. Ed., (2014), 53, 9560-9563
J. Am. Chem. Soc. (2010), 132, 2122 – 2123
Monolayer Systems
We created a new platform to employ in lipid monolayer and lipid membrane studies. Namely, a 3-dimensional (3D) phospholipid monolayer with tunable molecular structure was created on the surface of oil nanodroplets from a mixture of phospholipids, oil and water. Such a simple sample preparation technique generates an in-situ prepared membrane model system with controllable molecular surface properties. The molecular interfacial structure of such a nanoscopic system composed of hexadecane, 1,2-dihexadecanoyl-sn-glycero-3-phosphocholine (DPPC), and water was determined using vibrational sum frequency scattering and second harmonic scattering techniques. By varying the DPPC / oil/ water ratio, DPPC lipid structure can be tuned from a tightly packed liquid condensed phase like monolayer to a more dilute one that resembles the liquid condensed / liquid expanded coexistence phase. The tunability of the chemical structure, the high surface to volume ratio and the small sample volume make this system an ideal model membrane for biochemical research.
The system is very similar to lipid droplets that one finds as organels in almost every living cell.
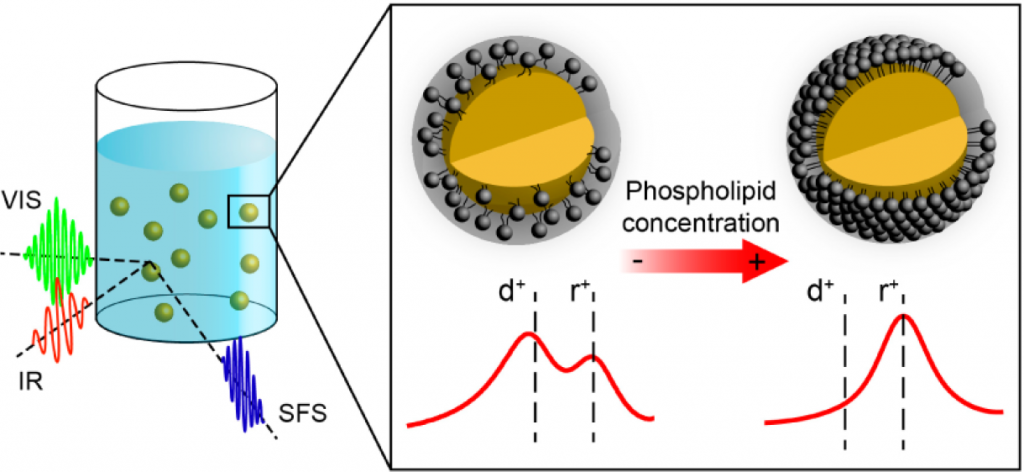
Nanodroplets with Lipid Monolayers. Left: Schematic showing the nanodroplets in aqueous solution in a sample cuvette. The red and green waves are representing the incoming IR and vis beams. The blue waves represent the generated sum frequency scattering beam. Right: Spectra showing the molecular configuration of the interface. Adapted from: Nano Lett. (2015), 15, (8), pp5558-5563
Liposomes
Variations between the inner and outer leaflets of cell membranes are crucial for cell functioning and signaling, drug–membrane interactions, and the formation of lipid domains. Transmembrane asymmetry can in principle be comprised of an asymmetric charge distribution, differences in hydration, specific headgroup / H-bonding interactions, or a difference in the number of lipids per leaflet. Here, we characterize the transmembrane asymmetry of small unilamellar liposomes consisting of zwitterionic and charged lipids in aqueous solution using vibrational sum frequency scattering and second harmonic scattering, label-free methods, specifically sensitive to lipid and water asymmetries. For single component liposomes, transmembrane asymmetry is present for the charge distribution and lipid hydration, but the leaflets are not detectably asymmetric in terms of the number of lipids per leaflet, even though geometrical packing arguments would predict so. Such a lipid transmembrane asymmetry can, however, be induced in binary lipid mixtures under conditions that enable H-bonding interactions between phosphate and amine groups. In this case, the measured asymmetry consists of a different number of lipids in the outer and inner leaflet, a difference in transmembrane headgroup hydration, and a different headgroup orientation for the interacting phosphate groups.
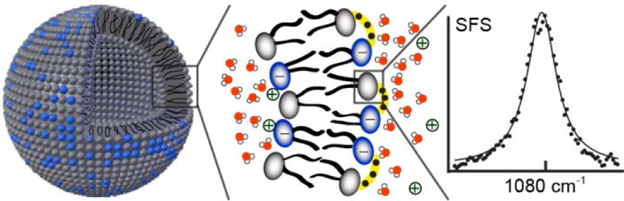
Transmembrane Asymmetry in Liposomes. Sum Frequency Scattering spectrum of H-bonded phosphate head groups on the outer leaflet of a mixed DOPC/DPPS liposome.From: JACS (2016),138 (12), pp. 4053-4060