The laboratory of biomedical microfluidics (LBMM) develops novel approaches for drug discovery and personalized cancer therapy. Having a strong translational focus, we collaborate with pharma industry, foster entrepreneurship and pave the way for first clinical trials. In line with this, we run a tech development lab on the EPFL campus, a translational lab in the AGORA cancer research center (with direct access to patient material) and we founded two startup companies making the results of our research widely available. The lab is very interdisciplinary and includes people with various backgrounds ranging from biology, genomics and bioinformatics to engineering and physics.
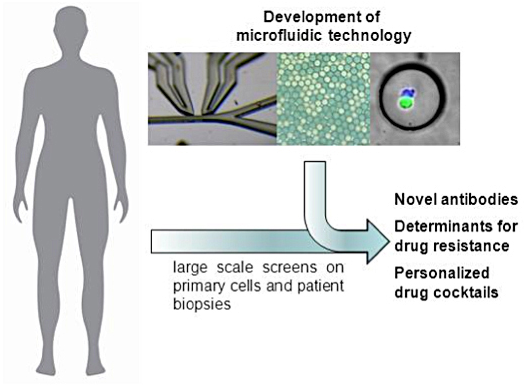
As a common theme we use microscopic droplets as miniaturized reaction vessels. This offers some unique advantages:
- Drastically increased throughput (processing up to a million samples an hour).
- Possibility of performing screens on the single-cell level (phenotypic and genotypic assays).
- Low sample consumption, enabling the use of patient material.
Having a comprehensive droplet microfluidic toolbox at hand (and expanding it continuously), we are now focusing on the following research themes:
Therapeutic antibodies and T-cells: Droplet based microfluidics enables to screen a large fraction of the murine and human immune repertoire in a single experiment (e.g. El Debs et al., PNAS 2012, Chaipan et al., Cell Chemical Biology 2017, Shembekar et al., Cell Reports 2018). We are actively exploiting this for novel therapeutic approaches and launched a first startup in 2017 (Velabs Therapeutics, now operating as www.veraxa.com), applying our technologies in real-world applications.
Personalised medicine: We use microfluidic devices to test drug combinations directly on solid human tumor samples and to predict optimal therapies (e.g. Eduati et al., Nature Communications 2018). Current efforts focus on the development of highly multiplexed genetic and imaging-based readouts. In parallel, we are constructing next generation instruments for first clinical trials and launched a second startup, making our personalized cancer therapy approaches available to the public (www.thera-me.ch).
Genomics: We are actively developing microfluidic modules for single-cell barcoding and sequencing. This will enable the study of the mechanisms of drug resistance in great detail. In the past we have also developed microfluidic modules for the shearing of genomic DNA into fragments that are short enough for next generation sequencing. This technology has been licensed and is now commercially available world wide, e.g. in form of the bioruptor (www.diagenode.com/en/p/bioruptor-one-sonication-device).
Microfluidic Technology Development: We are continuously developing microfluidic chips with improved functionality (e.g. fully automated droplet sorting and fusion, multiplexed-multicolor-multichannel sorting), design new hardware (e.g. for impedance measurements, temperature control, etc.) and program new control software (mostly LabVIEW & Python). The overarching goal is to make the technology more robust, more automated (= accessible to non-specialists), faster and more versatile.
Members of the group get a truly interdisciplinary education, which provides competitive advantages for various career paths. This is mirrored by the fact that lab alumni have become new leaders in academia, industry and startup companies.