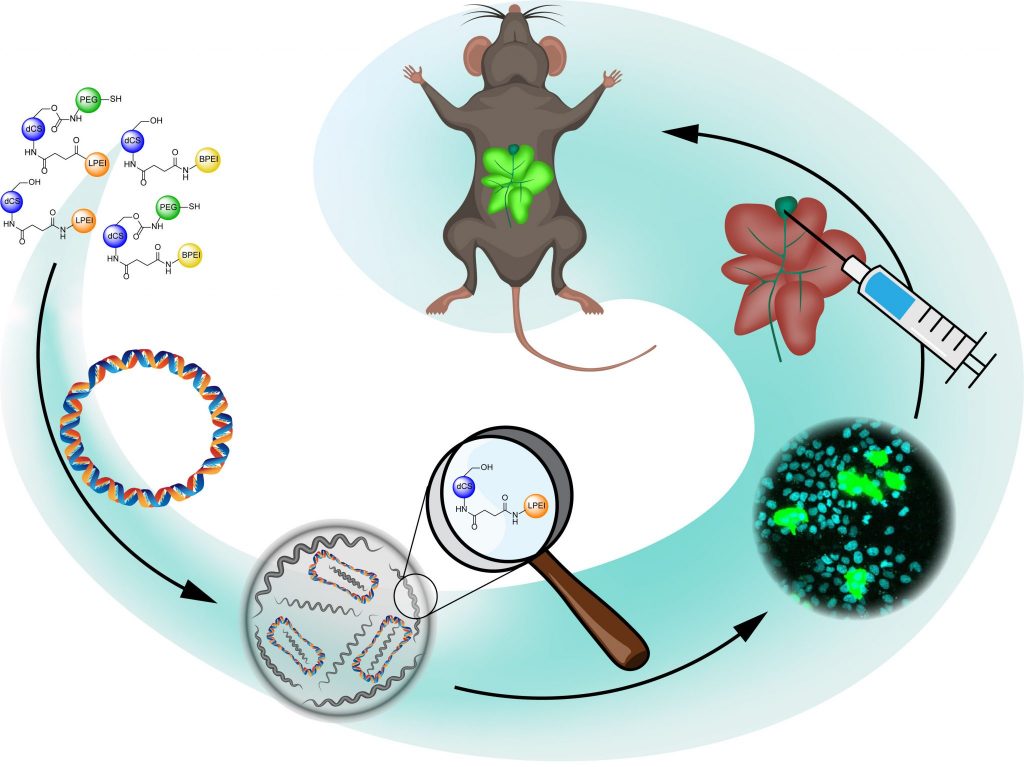
Development of polymeric chitosan-based materials for gene delivery
Gene therapy emerged in the last decades as a promising strategy for the treatment of acquired and inherited disorders, relying on the modification of gene expression by gene replacement or precise genome editing. Due to their polyanionic character and their sensitivity to endonuclease, nucleic acids should preferably be delivered to target cells by means of nanocarriers. Viral delivery vectors display high transfection efficiency but are hampered by inherent immunogenicity and potential cytotoxicity. As an alternative, synthetic non-viral vectors hold the potential for safer profile and easier production. In the frame of a Sinergia grant (UNIBASEL, EPFL, KISPI), we are currently developing polymeric gene delivery vehicles based on a chitosan (CS) backbone which is selectively functionalized with various molecular components promoting the key steps of DNA delivery: low molecular weight polyethylene imine (PEI) chains for efficient nucleic acid condensation, cell penetrating peptides to improve cellular uptake, cross-reactive PEG derivatives to stabilize the nanocarrier by cross-coupling, and NLS peptides to favour nuclear entry and transfection.
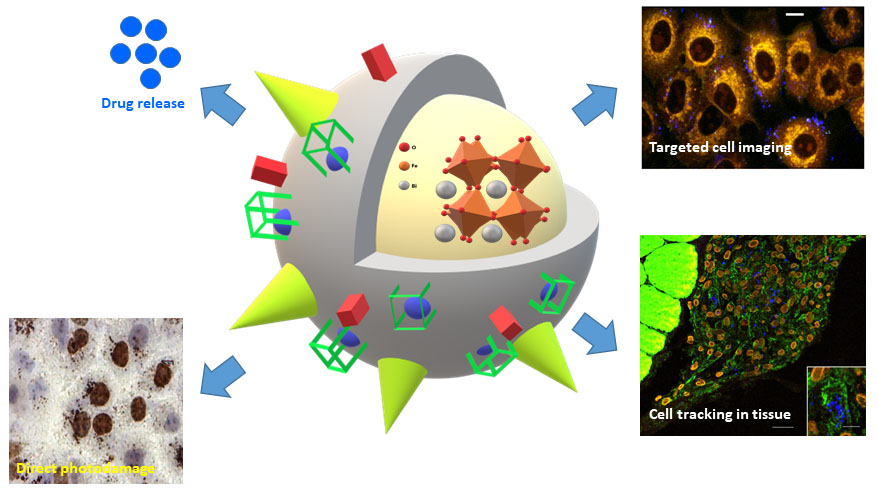
Surface modification of harmonic nanoparticles for bio-imaging and theranostic applications
Despite the recent progress in diagnosis and management of patients, cancer still represents a major health burden worldwide, in terms of morbidity and mortality. Among all cancer types, breast, lung and prostate cancer result in the highest social and economic impacts, each contributing to 12-13% of new cases and to high yearly death rates. One of the reasons for this situation lies in the insufficient level of sensitivity of the routine disease diagnostics at the very early stage of cancer development. The development of new diagnosis modalities pointing at early disease identification and prediction of metastasis development would have a major impact on survival rate and quality of life of patients. Harmonic nanoparticles (HNPs), which are increasingly perceived by the scientific community as complementary nanotools with respect to fluorescent/luminescent probes, present appealing properties for bioimaging applications, including long-term photostability, no-dependence on the value of excitation wavelength and narrow emission bands. In the frame of several collaborative projects (EPFL, UNIGE, SYMME-France, CHUV) aiming at the development of nanotechnological toolkits to provide qualitatively new levels of sensitivity and accuracy of malignant cells recognition in the context of lung, breat and prostate cancer, we developed synthetic methodologies for the biocompatible coating of HNPs and post-functionalization with targeting ligands addressing biomarkers of cancer cells and cancer-associated cells. In addition, we contributed to the demonstration of a novel scheme of HNP-mediated photo-interaction based on the emission at various wavelengths for direct photo-interaction with cellular DNA as localized therapeutic approach. In addition, a new detection protocol based on the multi-order nonlinear optical response of functionalized HNPs, particularly adapted to increase imaging selectivity for cell tracking in tissues, was established and further applied to stem cells tracking in tissue depth. In recent developments, we demonstrated the possibility to photo-induce the uncaging of small molecular cargos from the HNPs surface by the second harmonic generation of the metal oxide core, and the dual multiphoton imaging / MRI capabilities of HNP-based nanoconjugates presenting chelated Gd(III) ions at their surface. These achievements pave the way toward the development of versatile multifunctional nanoplatforms for multiscale multimodal imaging and targeted delivery of molecular and biomolecular payloads, which are currently investigated in our group.
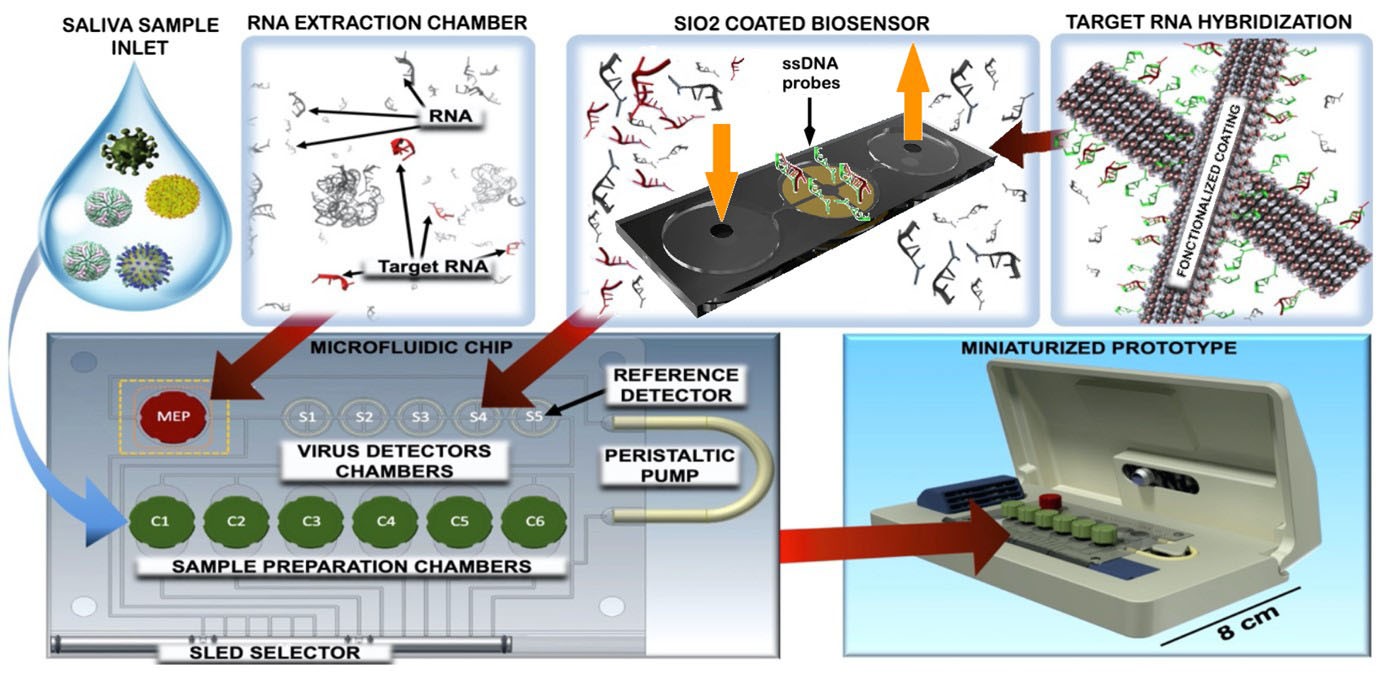
Development of portable sensing platforms for real time detection of viruses
Following the growing global outbreak of the Covid-19 pandemic, the scientific world has mobilised to rapidly produce solutions to reduce contagion and contain the spread of the novel coronavirus. Current techniques used for diagnosis include isolation of the virus in cell culture, genomic detection through Ribonucleic acid (RNA) amplification and serological testing (antigen and antibody detection). Current analysis methods imply a complex procedure, requiring hours for results, reagents and medical specialised personnel. The most relevant limit is related to the fact that this techniques are not deployable in airports, train stations, and ports, where the fast screening of travelling population would contribute to the early identification of potential vectors and would thus help in containing the disease propagation. In collaboration with the Department of Innovative Technologies at the University of Applied Sciences and Arts of Southern Switzerland (SUPSI), and the Institute of Oncology Research (IOR, affiliated to USI Università della Svizzera italiana), we aim at the development of an innovative diagnostic solution. The system will target viral RNA directly with an ultra-sensitive cheap biosensor made of porous silica (SiO2)-supported active sensing surfaces, aiming to reliably detect a minimum of 50 copies of viruses in 1 mL samples, in a handful of minutes. Our group is specifically exploring the different strategies for the production of the functionalized sensing surfaces. The technology will be applied to other types of viruses and will enable will enable the simultaneous detection of multiple pathogens.
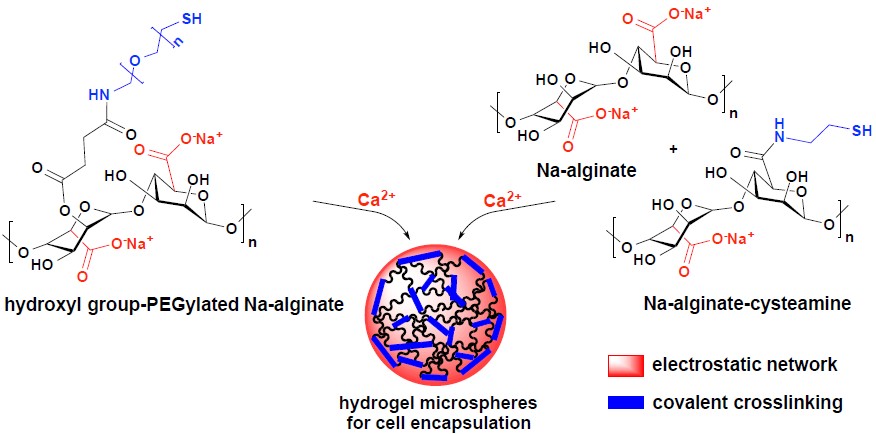
Development of functionalized polymeric hydrogels for cell immobilization
The increasing incidence of age-related diseases and the low availability of human donor material to replace dysfunctional cells and damaged tissues in patients have prompted the search for alternative transplantation therapies. Hydrogels, presenting a three dimensional (3D) structure, can serve as scaffolds for tissue engineering and as carrier for cell encapsulation and drug delivery. The transplantation of encapsulated cells/tissues prevents adverse immunological response while allowing the crossing of oxygen, nutrients and secreted factors. Despite the highly promising studies that have been directed toward the development of hydrogel microcapsules for the treatment of human diseases including liver failure, diabetes mellitus, Parkinson’s disease and anemia, routine clinical application of cell microcapsules still remains a challenge.
We develop synthetic routes for the production of PEG-grafted alginate hydrogels which are further conjugated to anti-inflammatory agents and drug scaffolds targeting fibrotic pathways to provide highly biocompatible materials for the encapsulation of islets and hepatocytes.
In vitro and in vivo evaluation of the resulting microcapsules, in collaboration with the University of Fribourg (Prof. Léo Bühler) address the long term functionality and viability of immobilized cells.