Epidural electrical stimulation (EES) of the spinal cord is a promising neuromodulation strategy that has been shown to facilitate locomotion in animal models of spinal cord injury (SCI) and of Parkinson’s disease (PD)1,2, and to a limited extent to humans patients with paraplegia3. Despite these promising results, the mechanisms through which EES facilitates motor control remain largely unknown, even though this understanding is essential for the translation of this paradigm into a viable clinical application.
In collaboration with the Courtine-lab, we work towards identifying these mechanisms by combining computational modelling and experimental procedures in rodents, non-human primates and human subjects4,5. Our final goal is to take advantage of the gained mechanistic understanding to optimize current EES strategies and to promote the clinical translation of this intervention.
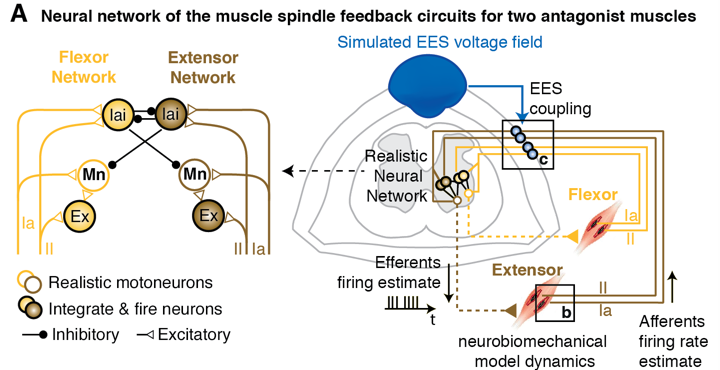
Mechanisms underlying species-specific responses to electrical spinal cord stimulation
We are currently studying the mechanisms underlying species-specific responses to EES in order to boost the limited efficacy of EES strategies developed in rodents for human applications. Using computer simulations, we showed that EES recruits the large proprioceptive afferents entering into the spinal cord from the dorsal roots, both in rodents and in humans. However, while we found that in rodents continuous EES increases the overall firing rate of the afferent fibers without disrupting the carried sensory information, in humans, simulations showed that EES interferes with the natural flow of sensory information. Consequently, we showed that continuous EES disrupts the modulation of proprioceptive feedback circuits in humans, strongly diminishing the facilitation of movements induced by this neuromodulation strategy. We are currently exploiting our computational framework to develop novel stimulation strategies that may remedy the disruption of sensorimotor circuits natural modulation, thus boosting the therapeutic efficacy of EES in humans.
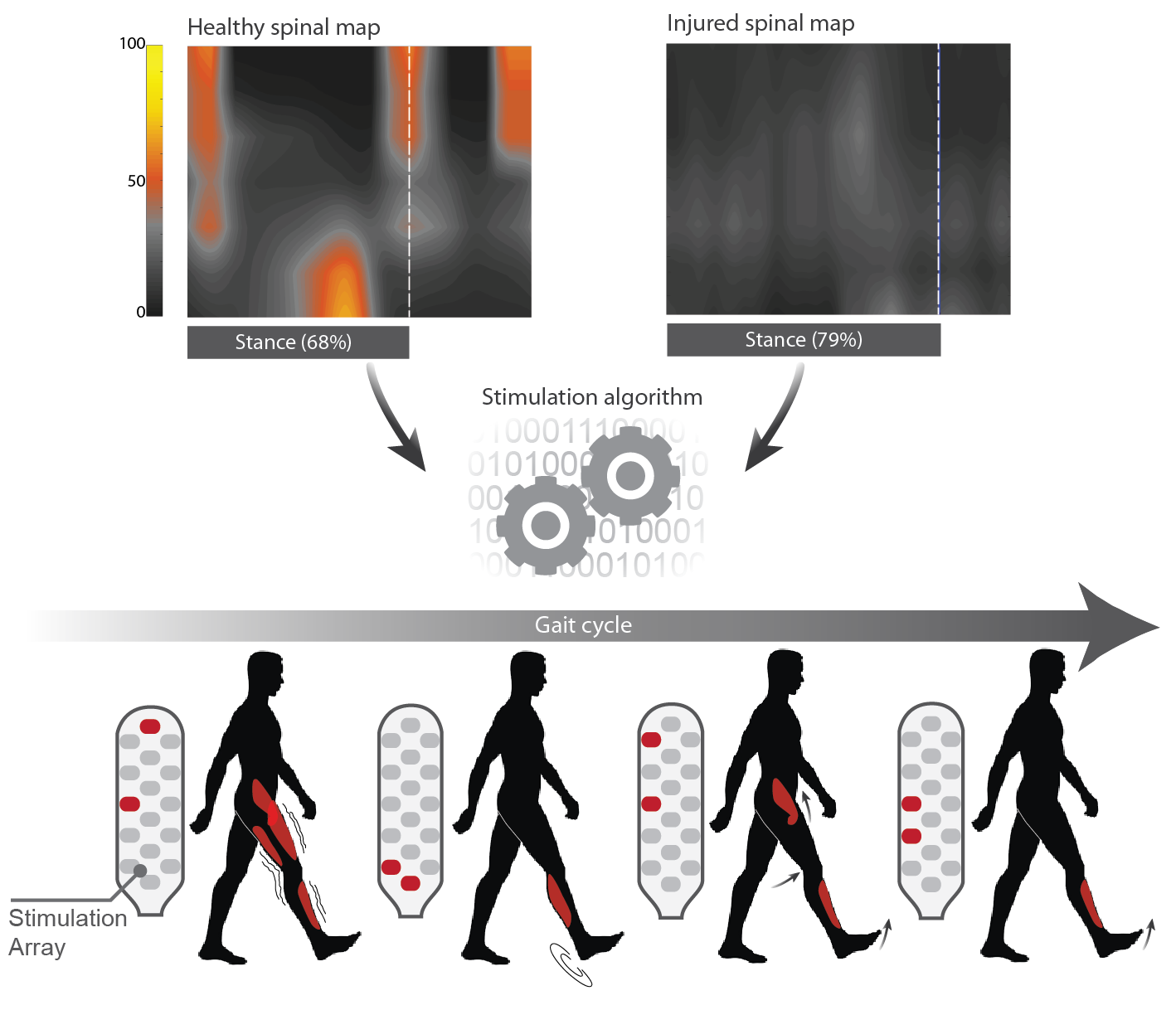
Subject-Dependent Spatiotemporal Electrical Neuromodulation Algorithm
Locomotion is a basic motor activity that requires the coordination of many limb and trunk muscles. Muscle activation is a reflection of α -motoneurons firing on the spinal cord. The activation of α -motoneurons during locomotor tasks seems to occur in bursts at discrete times, which depend on the speed and on limb loading. Recently, it has been shown in rats and monkeys that epidural electrical spinal cord stimulation (EES) can modulate the α -motoneuron activation and therefore replicate the healthy activation during locomotion.
To further reap the therapeutic benefits of spatiotemporal EES, it may be possible to improve the manner in which it is calibrated. The goal of the project is to design a subject-dependent spinal cord stimulation strategy based on α -motoneuron activation modulation to restore locomotor tasks in subjects with motor impairments.
The described algorithm is extremely versatile and translatable among diseases or injuries that cause locomotor disorders, such as spinal cord injury or Parkinson disease.
References:
- Van den Brand, R. et al. Restoring Voluntary Control of Locomotion after Paralyzing Spinal Cord Injury. Science 336, 1182–1185 (2012).
- Capogrosso, M. et al. A brain–spine interface alleviating gait deficits after spinal cord injury in primates. Nature Publishing Group 539, 284–288 (2016).
- Angeli, C. A., Edgerton, V. R., Gerasimenko, Y. P. & Harkema, S. J. Altering spinal cord excitability enables voluntary movements after chronic complete paralysis in humans. Brain 137, 1394–1409 (2014).
- Capogrosso, M. et al. A computational model for epidural electrical stimulation of spinal sensorimotor circuits. J. Neurosci. 33, 19326–19340 (2013).
- Moraud, E. M. et al. Mechanisms Underlying the Neuromodulation of Spinal Circuits for Correcting Gait and Balance Deficits after Spinal Cord Injury. Neuron 89, 814–828 (2016).
Contact
If you are interested in this research topic and wish to learn more, don’t hesitate to contact us:
Flavio Raschella ([email protected])