Elisa Oricchio- Curriculum Vitae
Cancer Genomics:
3D Structure, Gene Function, and Therapy
- The importance of study the cancer genome in 3D
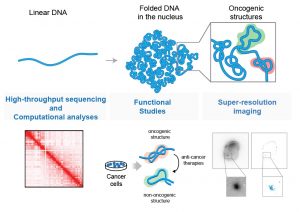
In the nucleus, the DNA is bounded with proteins and then, folded and looped in hierarchical structures. In cancer, genomic alterations modify the chromatin architecture to support tumor growth. A new area of research that I established in my lab integrates multiple computational, super-resolution imaging and functional approaches to understand the effect of cancer genomic alterations on chromatin structural elements (Figure 1). We recently completed a study, that investigates how recurrent mutations identified in several cancer patients can exploit the three-dimensional organization of the genome to sustain oncogenic programs (Donaldson-Collier et al 2018). With this approach, we are able to uncover unexplored oncogenic mechanisms, moving beyond the analysis of single genes but considering the complexity of the genome spatial organization.
Figure 1. Analysis of 3D structure of the chromatin in cancer cells
- Cancer genomics and personalized therapies
A better understanding of the effects of genomic alterations in tumorigenesis has steered cancer treatment towards more targeted and personalized therapies. However, curative approaches are rare and resistance to treatments and disease relapse are frequent and linked with tumor evolution. In my lab, we analyze genetic alterations, tumor heterogeneity, and oncogenic signals associated with lymphoma pathogenesis, to understand the evolution of the tumor and propose new therapies. To support the translational importance of our studies, we are taking advantage of patient derived xenograft (PDX) models and transgenic animal models. Recently, we analyzed response to targeted therapies in primary patient derived xenograft models and we uncovered new mechanisms of resistance to commonly used therapies and proposed an alternative therapeutic approach effective that could be effective for a broad group of patients (Oricchio et al. 2017, Battistello et al 2018).
Moreover, using primary patient derived xenograft models, we observed in vivo different patterns of tumor growth in response to different targeted therapies. Hence, we are using these models to study the effect of anti-cancer therapies on tumor evolution taking advantage of cutting edge single cells sequencing analyses.
Finally, in recent years, cancer treatment has been revolutionized by the introduction of immune therapies. For example, the use of engineered T-cells (CAR-T cells) has been recently approved for the treatment of patients with advanced lymphoma that are refractory to other therapies. However, as for targeted therapies, response to immunotherapies can be influenced by the tumor genetics. Hence, taking advantage of our experience in large scale genomic screen and our knowledge on the genetics of lymphoma, we are investigating the effects of genomic lesions on immune cells activity and how they influence the response to immunotherapies.
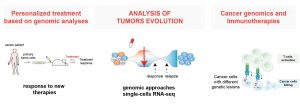
Figure 2: Overview of different approaches used in the lab to improve cancer therapies