The concept of cancer immunotherapy is an attractive approach to combine specificity and multi-dimensional targeting. Immunotherapy holds several advantages over conventional and targeted therapies. First, immunotherapy often results in better controllable side effects, enabling it to be administered for longer periods of time. And because of the immune system’s ability to target multiple cancer antigens simultaneously, patients may also be less likely to develop resistance.
Although breast cancer has historically been considered immunologically silent, several recent preclinical and clinical studies suggest that immunotherapy has the potential to improve outcomes for breast cancer patients. Immunotherapy development currently focuses on three treatment categories: vaccines, checkpoint blockade, and chimeric antigen receptor (CAR) T-cell therapies. The most advanced studies have used vaccination which improved the 5-year rate of disease-free survival (DFS) from 80 to 94%. Immune checkpoint inhibitors, which interfere with immune suppression against tumor-reactive immune cells, have shown striking results that have generated an enormous amount of excitement. Checkpoint blockade has been evaluated in metastatic triple-negative breast cancer (TNBC) patients and produced an overall response rate of 18.5%. Given that TNBC respond so poorly to all existing treatments, this is a significant progress. However, this also shows that still a majority of patients does not show benefit from this type of treatment indicating that available treatments may miss several important mechanisms of immune evasion.
One aspect which has not been sufficiently addressed is how different subpopulations of cancer cells and in particular how cancer stem cells (CSC) respond to immunotherapy. CSC have the unique ability to perpetuate tumor growth and heterogeneity and to initiate metastasis formation. Since it was shown that CSCs can be the source of recurrence, it will be highly relevant to study whether these cells can be targeted by immunotherapy.
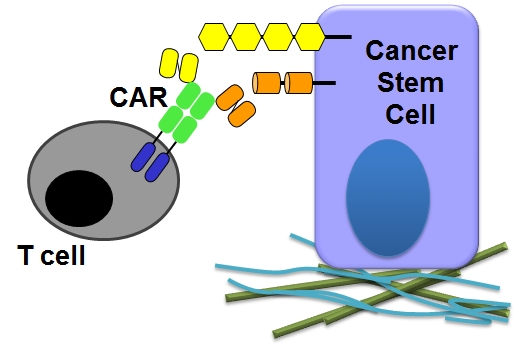
Genetic engineering of CD8+ T cells to express chimeric antigen receptors (CAR) will enable
specific elimination of the important cancer stem cell (CSC) population in tumors
CAR T cell therapies allow bypassing the MHC-restriction of the TCR by using antigen binding domains derived from antibodies. Such CARs contain the single chain fragment of variable regions (scFv) at the extracellular part plus the CD3zeta signaling domain of the TCR within the intracellular region. T-cells that express the CAR obtain a new specificity against a given antigen and signal through the CD3zeta domain which leads in the case of cytolytic T-cells (CTL) to the elimination of antigen-positive target cells. In contrast, antigen-negative target cells are not affected by these engineered T cells. Given the importance of CSCs for cancer formation, it will be also very important to test the possibility to specifically eliminate this important cancer ell subpopulation; possibly not only for treating the primary tumor but also to prevent metastatic progression.