Metastasis formation is cause of cancer-related death in over 90% of patients. However, our knowledge on how metastases initiate and grow is still rather limited. Surprisingly, it has been observed in experimetal models that metastasis formation is a rather inefficient, multi-step process with only very few cancer cells seeding growth in a distant organ.
Inefficient metastatic colonization is believed to reflect the inappropriate communication between tumor cells and the local microenvironment at a distant location in contrast to the primary site in which the tumor has initially evolved. Only a few disseminated tumor cells might be able to cope with the new environment. This is reflected in experimental metastasis models where repeated passages of tumors have been used to select for cell clones with increasing metastatic potential.
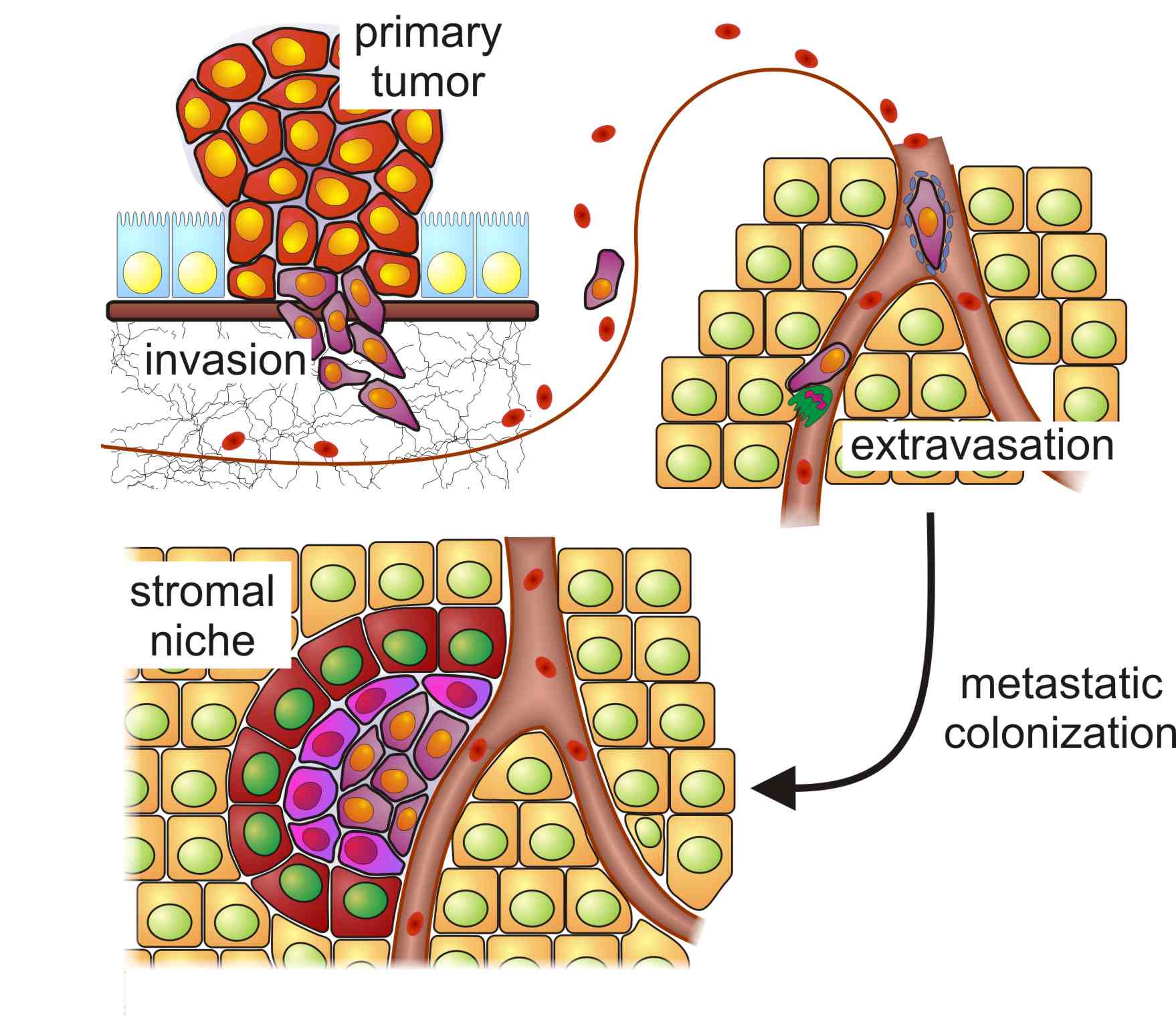
Important steps during metastasis formation: The metastatic cascade is initiated in the primary tumor by cells which gain migratory properties and invade into the surrounding stroma and the supplying vasculature. At the target organ, the cells actively extravasate or become trapped within the capillary network. Most of these cells will not be able to activate the surrounding stroma and either persist in a dormant state or die. Only very few tumor cells manage to adapt to the new environment, to activate the stroma and to form a micrometastasis. Few of these are able to expand after attraction of additional blood supply and grow into macrometastases (not shown).
We utilize an in vivo, murine model system of liver metastasis, which is the major metastatic target site for a variety of human cancers such as colon, pancreatic, gastric or ovarian cancers. This model enables us to study early events in metastatic initiation and outgrowth which cannot be analyzed in patients. In this experimental setting all metastases are initiated by identical oncogenic precursor cells; therefore differences in successful metastatic progression are due to specific interactions between tumor and local host cells. In vivo selection by the microenvironment in the target organ will favor certain combinations of tumor and host cell features which are able to support metastatic colonization to varying extent.
Together with the bioinformatics group of Felix Naef, we have developed a novel method to simultaneously analyze the changes which occur in the human tumor cells as well as the surrounding murine stroma. This approach relies on transcriptional profiling of chimeric tissues and allows us to analyze tissue-specific responses. Measuring gene expression levels of the tumor (human) and immediately adjacent stroma (mouse) without physically separating mouse and human cells avoids any degradation or expression changes which could occur during standard isolation procedures. This allows us to identify essential, reciprocal interactions at the tumor/host interface during metastatic colonization which will provide new possibilities of therapy.