SEMESTER PROJECTS/LAB IMMERSIONS 2025-2026
Regulation of metastatic dormancy in hormone receptor-positive breast cancer
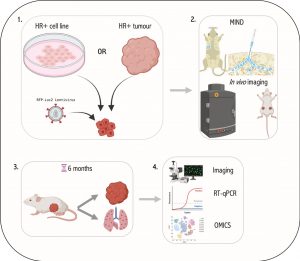
Breast cancers (BC) are divided into 4 main subtypes based on their expression of the oestrogen receptor (ER), progesterone receptor (PR) and the Human epidermal growth factor receptor (HER2) (1). ER+ breast cancer patients’ probability of survival decreases 5 – 20 years post-diagnosis due to minimal residual disease and metastatic relapse. This deadly phenomenon is caused by dormant DCCs at metastatic sites that remain undetectable at the time of initial diagnosis until giving rise to overt metastatic disease (2,3). Understanding the kinetics and molecular programmes employed by these DCCs during disease progression is essential for early intervention and prevention of this metastatic latency. In line with metastatic latency in ER+ breast cancer, it has been shown that metastatic cells from low burden tissues (HR+) displayed high expression of epithelial-to-mesenchymal transition (EMT), stem cell and dormancy genes compared to that of high burden tissues (HR-) (4,5). This suggests that cancer cells may undergo EMT and disseminate at early stages of tumour formation and seed as quiescent/dormant cells at metastatic sites for years before initiating secondary disease (6,7). As HR+ breast cancers make up roughly 70% of all breast cancers, late metastatic disease and cancer cell dormancy remains a significant clinical problem that has largely been overlooked by breast cancer researchers. This project aims to identify global regulators of EMT and metastatic dormancy in HR+ breast cancers using the MIND-model.
The student will gain experience with in vitro cell culture, ex vivo models and molecular biology techniques to tackle these questions.
References:
- Koboldt DC, Fulton RS, McLellan MD, Schmidt H, Kalicki-Veizer J, McMichael JF, et al. Comprehensive molecular portraits of human breast tumours. Nature. 2012;490(7418):61–70.
- Gawrzak S, Rinaldi L, Gregorio S, Arenas EJ, Salvador F, Urosevic J, et al. MSK1 regulates luminal cell differentiation and metastatic dormancy in ER + breast cancer. Nat Cell Biol [Internet]. 2018;20(2):211–21. Available from: http://dx.doi.org/10.1038/s41556-017-0021-z
- Zhang XHF, Giuliano M, Trivedi M V., Schiff R, Kent Osborne C. Metastasis dormancy in estrogen receptor-positive breast cancer. Clin Cancer Res. 2013;19(23):6389–97.
- Lawson DA, Bhakta NR, Kessenbrock K, Prummel KD, Yu Y, Takai K, et al. Single-cell analysis reveals a stem-cell program in human metastatic breast cancer cells. Nature. 2015;526(7571):131–5.
- Aouad P, Zhang Y, Stibolt C, Mani SA, Sflomos G, Brisken C. Epithelial-mesenchymal plasticity determines estrogen receptor positive (ER+) breast cancer dormancy and reacquisition of an epithelial state drives awakening. bioRxiv [Internet]. 2021 Jan1;2021.07.22.453458. Available from: http://biorxiv.org/content/early/2021/07/26/2021.07.22.453458.abstract
- Hosseini H, Obradovic MMS, Hoffmann M, Harper KL, Sosa MS, Werner-Klein M, et al. Early dissemination seeds metastasis in breast cancer. Nature [Internet]. 2016;540(7634):552–8. Available from: http://dx.doi.org/10.1038/nature20785
- Jiang J, Zheng M, Zhang M, Yang X, Li L, Wang S-S, et al. PRRX1 Regulates Cellular Phenotype Plasticity and Dormancy of Head and Neck Squamous Cell Carcinoma Through miR-642b-3p. Neoplasia. 2019 Feb;21(2):216–29.
Interested students are welcome to contact : [email protected]
Optimizing an ex vivo assay for testing hormone and drug response in hormone-sensitive breast cancer
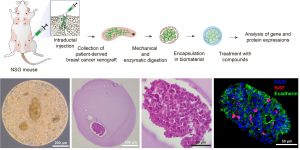
Breast cancer is a leading cause of cancer-related mortality for women worldwide (1). Endocrine therapy is of significant therapeutic value for hormone receptor-positive breast tumors that represent 70% of breast cancer cases (2). However, current standard endocrine therapies are largely based on pathological breast cancer features, and the outcomes are consequently affected by patient-to-patient heterogeneity. To provide tailored endocrine therapy, we propose a biomaterial-based 3D system to test hormone and drug sensitivity of patient-derived xenografts and patient tumor biopsies. The student will learn to manipulate and encapsulate tumors in biomaterials, and assess gene and protein expression using molecular biology and immunohistochemistry techniques, respectively.
References:
- Friedenreich CM. Physical activity and breast cancer: Review of the epidemiologic evidence and biologic mechanisms. Vol. 188, Recent Results in Cancer Research. Springer, Berlin, Heidelberg; 2011. p. 125–39.
- Harbeck N, Gnant M. Breast cancer. Vol. 389, The Lancet. 2017
Interested students are welcome to contact : [email protected]
Beyond Estrogen: The Overlooked Role of Androgens in normal breast epithelium and breast cancer
Background: Estrogen receptor-positive (ER+) breast cancer (BC) accounts for 70% of diagnosed cases, with treatments targeting ER including Tamoxifen, Fulvestrant, and aromatase inhibitors. However, the roles of progesterone receptor (PR) and androgen receptor (AR) in disease progression and treatment remain debated. Epidemiological studies link estrogen-androgenic progestin exposure to increased ER+ BC risk, suggesting AR involvement, though experimental results remain inconsistent. This study investigates AR signaling in normal human breast epithelium, an underexplored area.
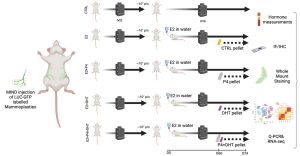
Using the Mammary IntraDuctal (MIND) model, we graft patient-derived tumor cells into mouse milk ducts, accurately replicating disease progression. Prior studies in our lab show that estrogen and progesterone act on “sensor cells”—ER+/PR+ luminal epithelial cells—through intrinsic and paracrine mechanisms. Progesterone induces two proliferation waves: an initial cyclin D1-dependent wave in sensor cells, followed by a broader, RANKL-mediated wave in receptor-negative mammary epithelial cells.
Rationale: There is an underappreciated role of androgen receptors (AR), both independently and in conjunction with estrogen (ER) and progesterone receptors (PR), in normal breast epithelium. We propose that testosterone collaborates with estrogen and progesterone to regulate breast epithelial dynamics during the menstrual cycle, driving cell growth, stem cell activation, and extracellular matrix remodeling—effects previously attributed solely to estrogen and progesterone in the luteal phase. Further, epidemiological and experimental evidence suggests a key distinction between androgenic progestins and natural progesterone in breast cancer risk, particularly in combined hormone replacement therapy (HRT) or elevated endogenous androgen levels. My work is to elaborate the effects of AR signaling in the context of normal breast epithelium and breast cancer development.
Specific aims:
1. Testing the hypothesize that testosterone acts as an enabler/amplifier of ER and PR signaling under physiologic conditions.
2. Test whether chronic exposure to an androgenic progestin but not an antiandrogenic progestin promotes early breast carcinogenesis.
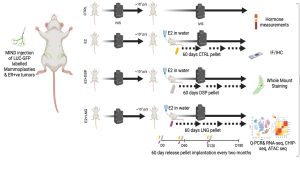
The student would work with patient samples, patient derived xenografts and observe mice work closely. The techniques learnt during training includes molecular and cell biology techniques like Western blotting, Q-PCR, Immunofluorescence, IHC, FACS, etc. and their respective analysis and data representation. Highly motivated students will also have a chance to do some exploratory studies that might contribute to the project.
Important references:
- Scabia V, Ayyanan A, De Martino F, Agnoletto A, Battista L, Laszlo C, et al. Estrogen receptor positive breast cancers have patient specific hormone sensitivities and rely on progesterone receptor. Nat Commun. 2022;13:3127.
- Shamseddin M, De Martino F, Constantin C, Scabia V, Lancelot A-S, Laszlo C, et al. Contraceptive progestins with androgenic properties stimulate breast epithelial cell proliferation. EMBO Mol Med. 2021;13:e14314.
Interested students are welcome to contact : [email protected]