Research

Our research focuses on the initial molecular events that launch an innate immune response. We are in particular interested in understanding how the immune system detects the presence of infectious viruses and how it responds to the emergence of cancerous cells. To dissect the fundamental mechanisms of innate immunity, we use all tools of modern biology, biochemistry, structural biology, and preclinical in vivo models of disease. On the applied side, we seek to harness insight into the functioning of the innate immune system to develop new paradigms for innovative medicines.
TO UNCOVER FUNDAMENTAL PRINCIPALS OF INNATE (DNA) IMMUNITY AND TO ELUCIDATE THEIR CONTRIBUTION TO HUMAN HEALTH AND DISEASE
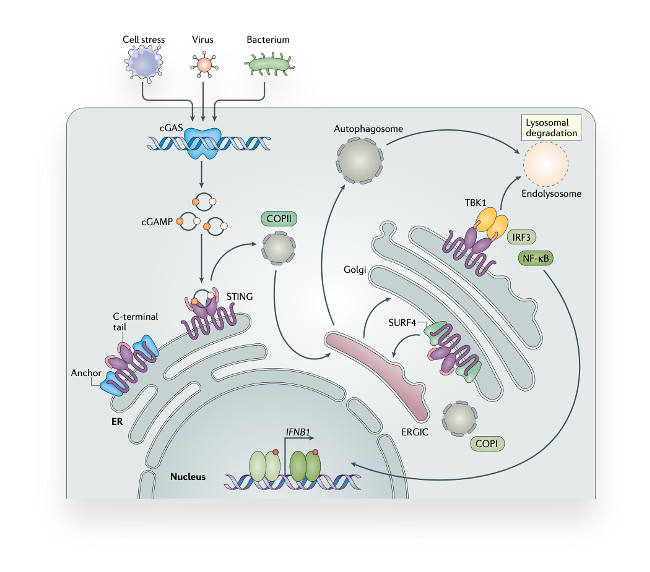
Sensing DNA as a danger signal through the cGAS-STING signaling pathway
The paradigm that DNA is a key initiator of inflammation was known even before it was shown that it serves as genetic material. However, how cells actually sense DNA from the inside and how they launch an inflammatory program was not understood until very recently. Today, we know that the cGAS-STING pathway recognizes DNA as a signal of infection or cell stress and, in turn, activates an innate immune response. Further, beyond infection, cGAS-STING signaling is associated with a range of distinct disorders, making it a highly attracted drug target. Still, many aspects about this highly conserved mechanism of innate immunity remain unknown.
(1) Ablasser, A., M. Goldeck, T. Cavlar, T. Deimling, G. Witte, I. Rohl, K. P. Hopfner, J. Ludwig and V. Hornung (2013). “cGAS produces a 2′-5′-linked cyclic dinucleotide second messenger that activates STING.” Nature.
(2) Ablasser, A., J. L. Schmid-Burgk, I. Hemmerling, G. L. Horvath, T. Schmidt, E. Latz and V. Hornung (2013). “Cell intrinsic immunity spreads to bystander cells via the intercellular transfer of cGAMP.” Nature.
(3) Ablasser, A. and Z. J. Chen (2019). “cGAS in action: Expanding roles in immunity and inflammation.” Science.
(4) Decout, A., J. D. Katz, S. Venkatraman and A. Ablasser (2021). “The cGAS–STING pathway as a therapeutic target in inflammatory diseases.” Nature Reviews Immunology.
Molecular rules that govern cGAS-STING pathway activity
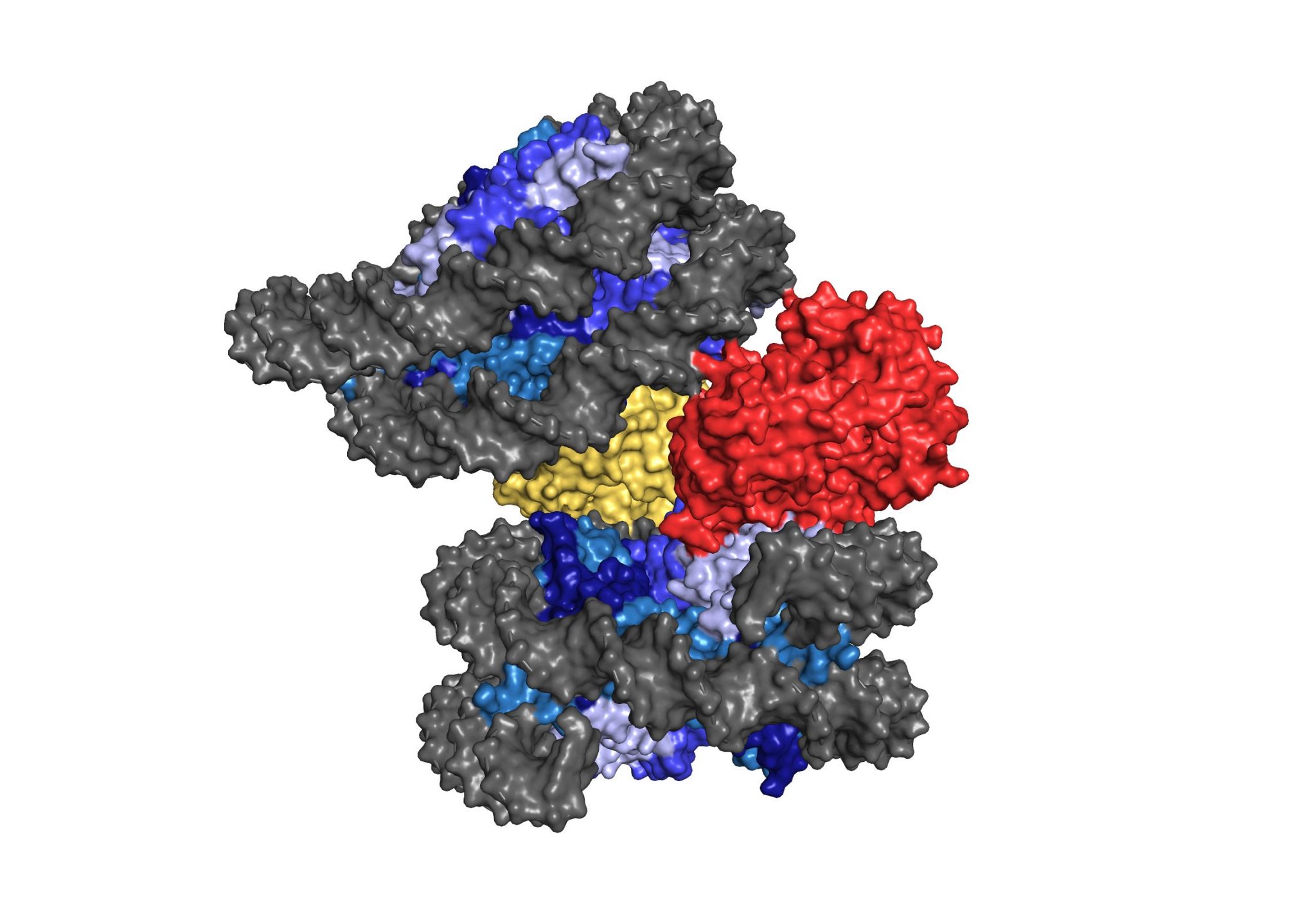
Immunity demands precision. Innate immune sensing mechanisms must be able to reliably sense infection or damage, and their function must be tightly controlled to achieve transient immune responses. We are interested in understanding regulatory principles that balance the activity of the cGAS-STING pathway at steady state and following activation. In prior work, we have elucidated fundamental mechanisms that safeguard the DNA sensor cGAS from responding to genomic DNA. We have identified barrier-to-autointegration factor 1 (BAF), as an essential chromatin architectural protein, that functions by displacing cGAS from nucleosome-free DNA regions to prevent autoreactivity. In addition, we have determined the cryo-electron microscopy structure of cGAS bound to the nucleosome revealing a structural mechanism of cGAS inhibition by host-derived chromatin.
cGAS-nucleosome structure (From Pathare et al.)
(1) Guey, B. and A. Ablasser (2022). “Interplay of cGAS with chromatin” Emerging dimensions of cellular cGAS-STING signaling” Current Opinion in Immunology.
(2) Wischnewski, M. and A. Ablasser (2021).”Interplay of cGAS with chromatin” Trends in Biochemical Sciences.
(3) Ablasser, A. and S. Hur (2020). “Regulation of cGAS- and RLR-mediated immunity to nucleic acids.” Nat Immunol.
(4) Guey, B., M. Wischnewski, A. Decout, K. Makasheva, M. Kaynak, M. S. Sakar, B. Fierz and A. Ablasser (2020). “BAF restricts cGAS on nuclear DNA to prevent innate immune activation.” Science.
(5) Pathare, G. R., A. Decout, S. Glück, S. Cavadini, K. Makasheva, R. Hovius, G. Kempf, J. Weiss, Z. Kozicka, B. Guey, P. Melenec, B. Fierz, N. H. Thomä and A. Ablasser (2020). “Structural mechanism of cGAS inhibition by the nucleosome.” Nature.
Drug discovery efforts harnessing the cGAS-STING pathway
Research pursued by many laboratories demonstrated that abnormal activation of the cGAS-cGAMP-STING signaling pathway is associated with a number of severe autoinflammatory diseases, including neurodegenerative diseases, cancer, and cardiovascular diseases. Therefore, pharmacological blockade of the cGAS-STING pathway is a highly attractive novel strategy harnessed to treat inflammatory conditions more efficiently. In prior work, we have identified small-molecule antagonists of STING and
Comment on STING inhibitors characterized an unexpected mechanism of pharmacological inhibition of STING. Our work described the first reported STING inhibitors and, using these compounds, we have provided proof-of-concept studies that STING inhibitors can alleviate autoinflammatory diseases in vivo. The finding that STING is druggable excited huge interest from the pharmacological industry and led to the foundation of IFM due, a subsidiary of IFM Therapeutics, that is developing cGAS- and STING targeting drugs for the treatment of inflammatory disorders. We have also defined apoptosis as a prime outcome of the cGAS-STING pathway, which will need to be considered when designing cGAS-STING targeting pharmaceuticals.

(1) Haag, S. M., M. F. Gulen, L. Reymond, A. Gibelin, L. Abrami, A. Decout, M. Heymann, F. G. V. Goot, G. Turcatti, R. Behrendt and A. Ablasser (2018). “Targeting STING with covalent small-molecule inhibitors.” Nature.
(2) Gulen, M. F., U. Koch, S. M. Haag, F. Schuler, L. Apetoh, A. Villunger, F. Radtke and A. Ablasser (2017). “Signalling strength determines proapoptotic functions of STING.” Nat Commun.
(3) Decout, A., J. D. Katz, S. Venkatraman and A. Ablasser (2021). “The cGAS–STING pathway as a therapeutic target in inflammatory diseases.” Nature Reviews Immunology.
Elucidation of the relevance of the cGAS-STING pathway in infection and disease
A major goal of our work is understanding the relevance of the cGAS-STING pathway in contexts of infection and pathology and to develop novel therapeutic paradigms. In earlier work, we have shown that inside macrophages cGAS is an important pattern recognition receptor for detecting Mycobacterium tuberculosis and for regulating type I interferon production and other regulatory cytokines, which have detrimental consequences in tuberculosis. We have also demonstrated that the cGAS-STING pathway is a prime mechanism by which self-DNA is controlling the inflammatory output of senescent cells – allowing for the removal of damaged cells by the immune system. In most recent work, we have demonstrated that the cGAS-STING pathway is engaged during SARS-CoV-2 infection and a major driver underlying type I IFN immunopathology.
(1) Wassermann, R., M. F. Gulen, C. Sala, S. G. Perin, Y. Lou, J. Rybniker, J. L. Schmid-Burgk, T. Schmidt, V. Hornung, S. T. Cole and A. Ablasser (2015). “Mycobacterium tuberculosis Differentially Activates cGAS- and Inflammasome-Dependent Intracellular Immune Responses through ESX-1.” Cell Host Microbe.
(2) Gluck, S., B. Guey, M. F. Gulen, K. Wolter, T. W. Kang, N. A. Schmacke, A. Bridgeman, J. Rehwinkel, L. Zender and A. Ablasser (2017). “Innate immune sensing of cytosolic chromatin fragments through cGAS promotes senescence.” Nat Cell Biol.
(3) Di Domizio, J., M. F. Gulen, F. Saidoune, V. V. Thacker, A. Yatim, K. Sharma, T. Nass, E. Guenova, M. Schaller, C. Conrad, C. Goepfert, L. De Leval, C. von Garnier, S. Berezowska, A. Dubois, M. Gilliet and A. Ablasser (2022). “The cGAS-STING pathway drives type I IFN immunopathology in COVID-19.” Nature.