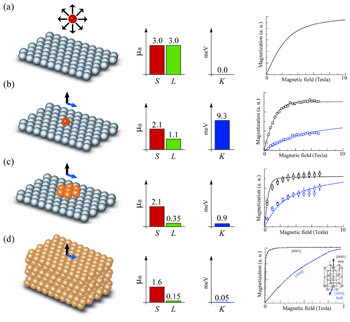
Knowing how the magnetic moment of a transition metal atom (a) change upon deposition on a nonmagnetic substrate (b) and bonding with neighbour atoms (c) are two basic steps to understand the evolution of the magnetization in supported nanoparticles and thin films. Due to the reduction of spatial symmetry and coupling with the substrate, a free-atom develops a strong magnetic anisotropy energy (K) at an atomically ordered surface. K, the orbital (L) and spin (S) magnetic moment depend critically on the atomic coordination [P. Gambardella et al., Science 300, 1130 (2003)].
Introduction
There are good theoretical models and plenty of experimental data that tell us how magnetism works in free atoms (Fig. 1a) and crystalline solids (Fig. 1d). However, as we explore the realm in between these two limits (Fig. 1b-c), we face a remarkable paucity of experimental information. Most elements in the periodic table are magnetic as free atoms. Only iron, cobalt, nickel, a few rare-earths and their alloys exhibit ferromagnetic properties in bulk compounds. The evolution of the atomic magnetic moments, their mutual coupling which ultimately produces macroscopic magnetism, and the appearance of magnetic anisotropy in molecules, nanosized aggregates, and solids are some of the main issues investigated by our group. We use a combined experimental approach to relate the atomic-scale morphology of dilute alloys, nanoparticles, and wires to their magnetic properties. Besides being of fundamental interest, such questions bear on the design of novel magnetic devices with one or more dimensions reduced to the length scale of interatomic cooperative magnetic behavior. The most relevant technological applications include giant magnetoresistance sensors, currently used in the read-heads of computer hard disks, spin-based electronics, of great promise for integrated memory and electronic devices, and the conception of new magnetic media, soft and hard magnets.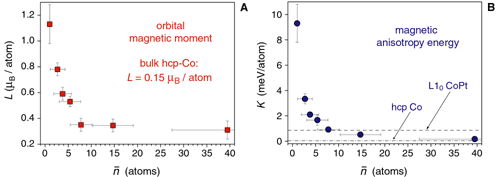
Fig. 2:
Size dependence of the magnetic anisotropy energy (K) and orbital magnetic moment (L) in two-dimensional Co nanoparticles on a Pt(111) surface [P. Gambardella et al., Science 300, 1130 (2003)].
Current Projects
- Magnetic impurities on nonmagnetic metal surfaces
- Magnetic behavior of transition metal layers, clusters, and nanowires
- Paramagnetic metal-organic complexes at surfaces
Experimental techniques
- Spin-polarized scanning tunneling microscopy (SP-STM) – see Fig. 3
- X-ray absorption spectroscopy (XAS) and x-ray magnetic circular dichroism (XMCD) – see Fig. 4
- X-ray photoemission electron microscopy (XPEEM)
- Angle-resolved ultraviolet photoemission spectroscopy (ARUPS).

Fig. 3:
Explosed view of the STM z-motor (Pan – Wiesendanger design) and scanning tip.
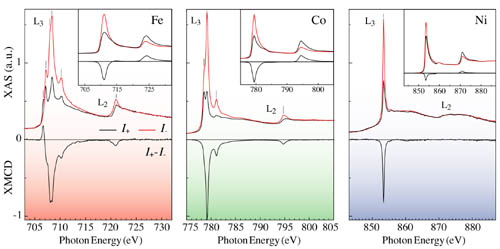
Fig. 3: X-ray absorption spectroscopy with synchrotron radiation allows to determine the valence state and magnetic moment of metal impurities with great sensitivity. The figure shows the XAS and XMCD spectra (L2,3 edges) recorded for isolated Fe, Co, and Ni impurities on a K surface corresponding to a coverage of less than 0.01 monolayers. Multiplet spectral features are due to electron localization at the impurity sites and disappear in the bulk metal spectra (insets). Measurements performed at the ID08 beamline of the European Synchrotron Radiation Facility in Grenoble. [P. Gambardella et al., Phys. Rev. Lett. 88, 047202 (2002)].