Biological systems require unique control of materials and interfaces to enable benefits to human health. We leverage our expertise in fabrication of complex microsystems to enable new applications in biology and medicine.
An Ingestible Bioprinter for Non-Invasive Structured Bioink Deposition

We present the first tetherless, ingestible bioprinter capable of controlled bioink deposition and patterning for potential applications in endoluminal tissue repair. This ingestible microdevice operates without tethers or onboard electronics and is guided using external imaging and magnetic actuation. By integrating near-infrared (NIR) triggering and magnetic guidance — both applied from outside the body — the device can navigate to target sites and deposit bioink in defined patterns, enabling ulcer coverage or hemorrhage sealing in ex vivo gastrointestinal tissue models.
A shape-memory crossbeam made from melanin-doped PLA contracts upon NIR irradiation, releasing a spring-loaded plunger that extrudes bioink through a gold-coated NdFeB ring magnet nozzle. Alginate-based, cell-laden constructs printed by the device retained 73% of alginate mass under simulated gastric conditions and remained viable for over 16 days, demonstrating potential as micro-bioreactors for sustained growth factor delivery and wound healing. In vivo rabbit studies confirmed fluoroscopy-guided positioning, controlled extrusion, and structured patterning — establishing the core engineering principles for future non-invasive endoluminal bioprinting systems.
Key highlights:
- First demonstration of a tetherless, electronics-free ingestible bioprinter
- NIR-triggered shape-memory extrusion mechanism with sub-second actuation response
- Ex vivo hemorrhage sealing and ulcer dressing using alginate bioink
- Cell-laden constructs viable for 16+ days; alginate retained under gastric simulation
- In vivo rabbit feasibility: fluoroscopic guidance, positioning, and bioink deposition
Publication
Manoharan S & Subramanian V. A Pill That Prints — An Ingestible Bioprinter for Non-Invasive Structured Bioink Deposition. Advanced Science, Vol. 12, Issue 46, e12411 (2025). https://doi.org/10.1002/advs.202512411
Affiliated members
Sanjay Manoharan
Magnetically-Guided, Stimuli-Responsive Microdevices for Endovascular Therapy
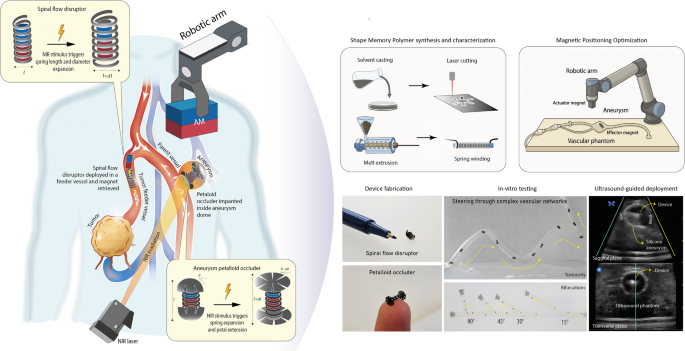
Current endovascular aneurysm treatments rely on catheter-based delivery systems that restrict access to tortuous anatomies and small-caliber vessels. We introduce a tetherless microdevice platform combining magnetic guidance with near-infrared (NIR)-triggered shape-memory polymer (SMP) deployment for wireless aneurysm therapy. An external actuator magnet steers a microdevice-integrated effector magnet through anatomically realistic silicone vascular phantoms, while melanin-doped PLA structures enable precise NIR-induced shape recovery activated non-invasively through the body wall.
The platform supports two device architectures tailored to different clinical needs: (1) a spiral flow disruptor with a retrievable magnet for partial inflow modulation, and (2) a petalloid occluder designed for permanent sealing of narrow-neck aneurysms. Navigation behaviour is modelled using a novel flow-responsive, magnetically modulated stick-slip (F-MMSS) framework capturing the influence of pulsatile flow and wall interactions. Magnetic steering was demonstrated through complex vascular networks, with final deployment validated under ultrasound guidance.
Key highlights:
- Tetherless platform: no catheter required; steered entirely by external magnetic actuation
- NIR-triggered shape-memory deployment (melanin-doped PLA) activatable through biological tissue
- Two device architectures: spiral flow disruptor (retrievable) and petalloid occluder (permanent)
- F-MMSS analytical model predicts navigation in pulsatile flow through tortuous vascular phantoms
- Ultrasound-guided deployment demonstrated in realistic silicone vascular models
Publication
Manoharan S & Subramanian V. Magnetically-Guided, Stimuli-Responsive Microdevices for Endovascular Therapy. Biomedical Microdevices, Vol. 28, Article 6 (2026). https://doi.org/10.1007/s10544-026-00791-4
Affiliated members
Sanjay Manoharan