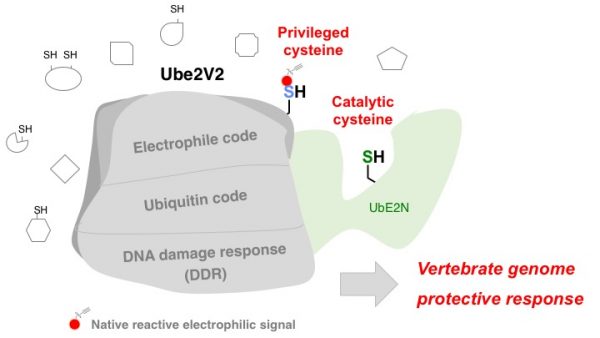
Ube2V2 is a Rosetta Stone bridging redox and ubiquitin codes, coordinating DNA damage responses
Synopsis: G-REX and T-REX double tap strategy identifies privileged electrophile responsivity of Ube2V2 to promote genome protection
Yi Zhao#, Marcus J. C. Long, Yiran Wang, Sheng Zhang, and Yimon Aye*
(#, first author. *, corresponding author)
Highlight in Cornell Chronicle
We thank all of the contributors in this team’s work: Yi is a former graduate student organic chemist of the National University of Singapore, now a senior postdoctoral colleague in the Aye Lab having been fully converted to chemical/molecular/cell biologist and zebrafish biochemist during the course of his postdoctoral training. Marcus is a senior postdoctoral colleague in the Aye Lab and a former graduate student biochemist of Brandeis University. Yiran is one of our dedicated former undergraduate researchers now pursuing her post-bac research internship at the NIH. Dr. Zhang is the Director of Cornell Proteomics facility. We also thank Nikki Gilbert and Brian Miller from Professor Joe Fetcho’s laboratory for assistance with zebrafish husbandry and breeding.
Abstract
Posttranslational modifications (PTMs) are the lingua franca of cellular communication. Most PTMs are enzyme-orchestrated. However, the reemergence of electrophilic drugs has ushered mining of unconventional/non-enzyme-catalyzed electrophile-signaling pathways. Despite the latest impetus toward harnessing kinetically- and functionally-privileged cysteines for electrophilic-drug design, identifying these sensors remains challenging. Herein, we designed “G-REX”—a technique that allows controlled release of reactive electrophiles in vivo. Mitigating toxicity/off-target effects associated with uncontrolled bolus exposure, G-REX tagged first-responding innate cysteines that bind electrophiles under true kcat/Km conditions. G-REX identified two allosteric ubiquitin-conjugating proteins—Ube2V1/Ube2V2—sharing a novel privileged-sensor-cysteine. This non-enzyme-catalyzed-PTM triggered responses specific to each protein. Thus, G-REX is an unbiased method to identify novel functional cysteines. Contrasting conventional active-site/off-active-site cysteine-modifications that regulate target activity, modification of Ube2V2 allosterically hyperactivated its enzymatically-active binding-partner Ube2N, promoting K63-linked client ubiquitination and stimulating H2AX-dependent DNA-damage-response (DDR). This work establishes Ube2V2 as a Rosetta-stone bridging redox and ubiquitin codes to guard genome integrity.
Aside: “… on July 19, 1799, the Rosetta Stone unlocked the language of the pharaohs.” (www.history.com)