In the broadest terms, our research interest is an amalgamation of chiral catalyst or ligand design and the development of new reaction methodology, which seeks to push frontiers of asymmetric catalysis by addressing longstanding limitations and critical problems in organic synthesis. Essentially, we aim to leverage on creative, direct and unique disconnections in important complex molecular framework, emphasizing on transition metal-catalyzed state-of-the-art C-H functionalization processes which enable asymmetric carbon-carbon and carbon-heteroatom bond forming transformations.
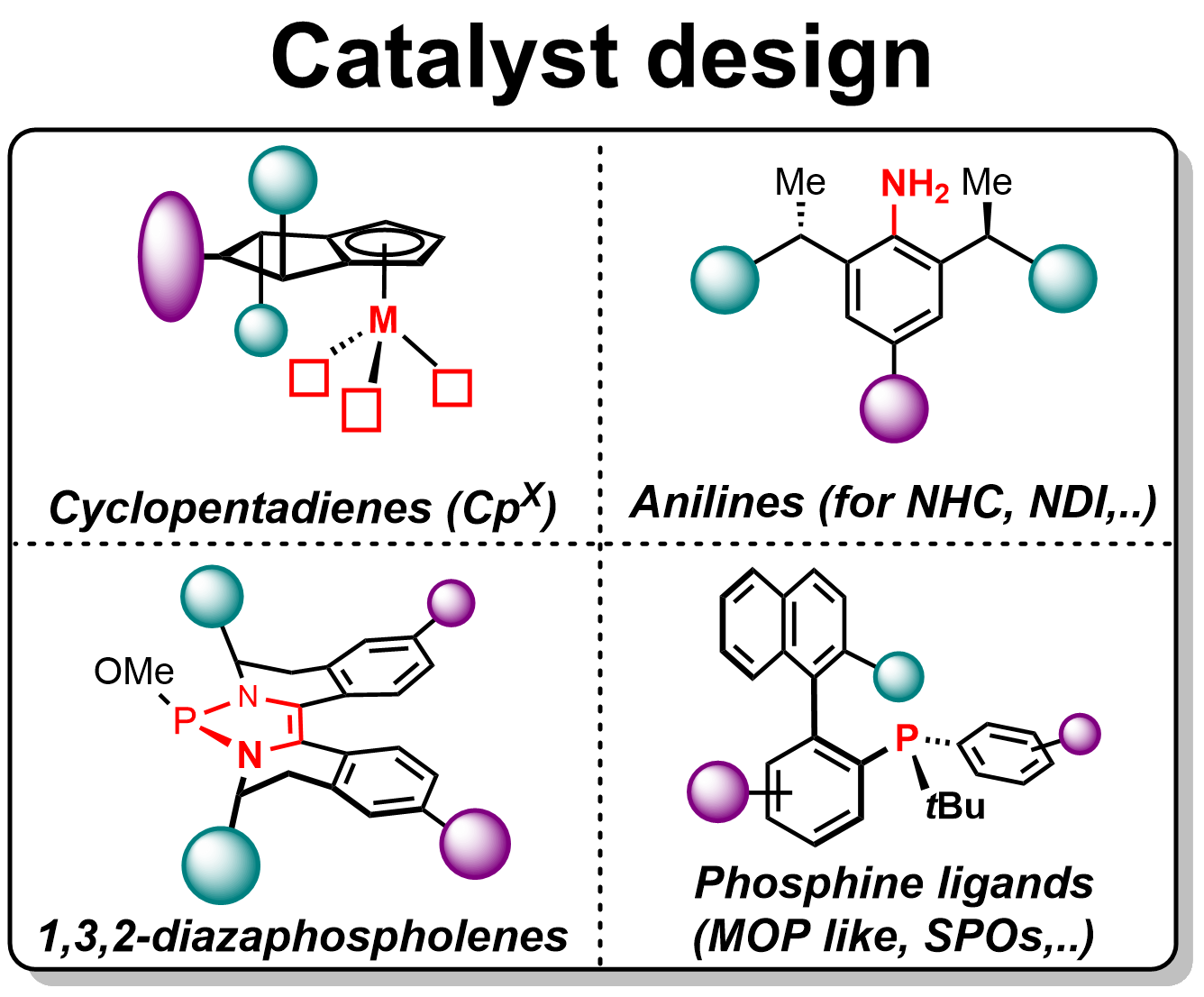
In our ongoing quest to develop novel asymmetric transformations, a variety of transition-metal catalysts are employed such as rhodium-, palladium-, iridium-, nickel-, cobalt-, and ruthenium-complexes. In particular, we currently focus on four main robust ligand families that fit the needs of these metals and the envisaged reaction mechanisms:
- Chiral cyclopentadienyl (Cpx) ligands (derived from BINOL, mannitol, fulvenes);
- N-heterocyclic carbenes (NHC), napththyridine diamine (NDI) ligands derived from novel chiral anilines;
- Monodentate or bidentate phosphine-based ligands (MOP, BINAP, TADDOL-derived);
- 1,3,2-Diazaphospholene ligands (main group chemistry).
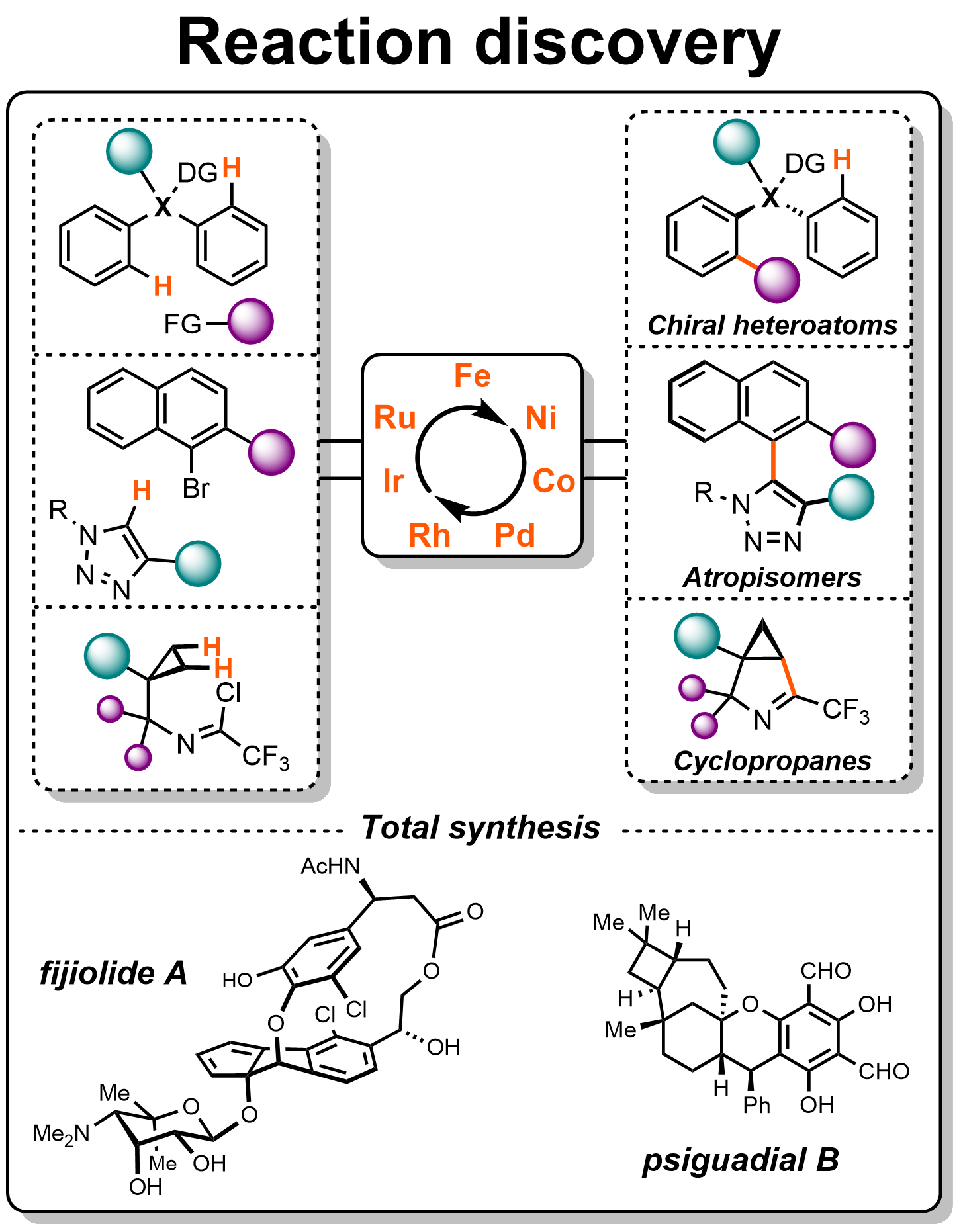
Our efforts in devising highly enantioselective reactions are fueled by insights gained from mechanistic studies, which assist us to continually improve and fine-tune our catalyst systems with the goal of establishing maximized efficiency and selectivity, while rapidly generating molecular complexities from simple, abundant and cost-efficient feedstocks. We apply these methods to synthesize bioactive molecules and complex natural products with intriguing pharmacological or agricultural properties.
For more details on our completed projects, please see our Publications section.