ADVANCED BIOENGINEERING METHODS LABORATORY (ABML)
Advanced Bioengineering Methods Laboratories (ABML) is part of the bioengineering master teaching courses and offers laboratory practice and classroom data analysis.
These active sessions present a variety of techniques employed in the bioengineering field and matching a quantitative and technological based approach. They promote the microtechnology role in life science community, mimic a real research project and focus on a couple of aspects such as integrating engineering approach, compiling proper lab notebook, respecting safe behavior as well as writing scientific report.
This page presents briefly the six practical exercises and makes available a number of useful documents and tools (i.e. the course program and description, practical handouts, matlab scripts for each analysis session, part of the references, video examples, lab notebook guidelines and an introduction to error computing). Students are asked for printing and studying handouts ( 6 topics + master handout) before each practical session.
- Introductory lecture 2017-2018.pdf
- BIO444 course book.pdf
- Error Propagation_2013.pdf
- AMBLError propagation 2013.pdf
To access directly to a specific section, click on it
Microfluidics Lab on a Chip
Brownian Motion
Optical Trapping
Surface Design
Surface Plasmon Resonance
Atomic Force Microscopy
Course Rules
Lab Notebook
Safety
Supplementary Materials
Contacts
Atomic Force Microscopy
 The main driving force behind the development of any microscope technique is the desire to look at things with ever higher resolution, ideally down to the atomic level. The Atomic Force Microscope (AFM) is one of the highest resolution microscopy techniques available, and the only one that can resolve atoms and single molecules of biological samples in aqueous solution. Therefore it has become an essential tool for nanoscale biology. It can be used for imaging single molecules, live cells or biological tissue with sub-nanometer resolution. In this lab, you will learn the basics of atomic force microscopy, image single molecules as well as study the quaternary structure of collagen.
The main driving force behind the development of any microscope technique is the desire to look at things with ever higher resolution, ideally down to the atomic level. The Atomic Force Microscope (AFM) is one of the highest resolution microscopy techniques available, and the only one that can resolve atoms and single molecules of biological samples in aqueous solution. Therefore it has become an essential tool for nanoscale biology. It can be used for imaging single molecules, live cells or biological tissue with sub-nanometer resolution. In this lab, you will learn the basics of atomic force microscopy, image single molecules as well as study the quaternary structure of collagen.
The AFM used for this exercise is one of the most advanced commercially available AFMs, and the only one in Switzerland. In the analysis section, you will learn how to extract quantitative data from the AFM images, use 3D-image reconstruction and advanced image processing.
Brownian Motion
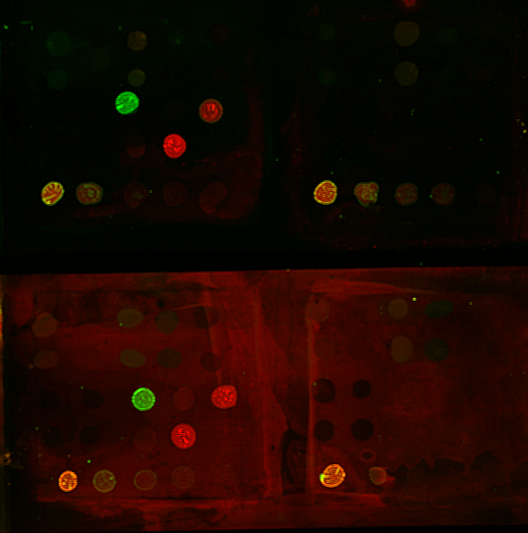
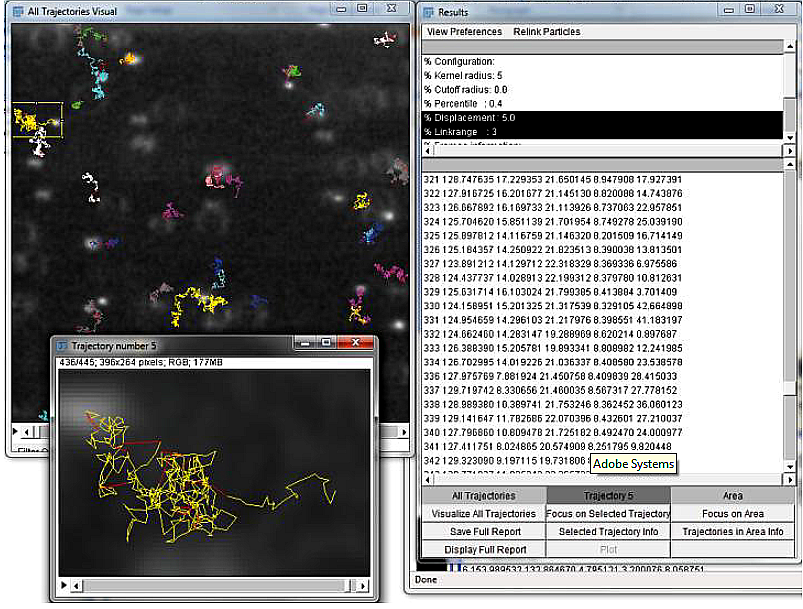 This lab begins with the ground-breaking physics of a century ago demonstrating the atomic nature of matter and ends with today’s biophysics state-of-the-art of intracellular transport and molecular motors. The pairing of advanced light microscopy with automated image analysis and particle tracking software provides a powerful tool for investigating the motion of molecules, organelles, and cells. In the first part of this lab, Perrin’s work will be replicated with such modern equipments: the motion of synthetic beads suspended in liquids of various viscosities will be tracked and studied. In the second part, the motion of particles inside living cells will be observed. Thereby this practical will introduce the bases of bead suspension sample preparation, microscopy aspects, particle detection and tracking as well as data analysis using matlab.
This lab begins with the ground-breaking physics of a century ago demonstrating the atomic nature of matter and ends with today’s biophysics state-of-the-art of intracellular transport and molecular motors. The pairing of advanced light microscopy with automated image analysis and particle tracking software provides a powerful tool for investigating the motion of molecules, organelles, and cells. In the first part of this lab, Perrin’s work will be replicated with such modern equipments: the motion of synthetic beads suspended in liquids of various viscosities will be tracked and studied. In the second part, the motion of particles inside living cells will be observed. Thereby this practical will introduce the bases of bead suspension sample preparation, microscopy aspects, particle detection and tracking as well as data analysis using matlab.
- Brownian motion in cells lecture
- Sbalzarini, Feature poit tracking and trajectory analysis for video imaging in cell biology, 2005
- Youtube Brownian Motion Examples
Microfluidics Lab on Chip
Recent progress in reconstructing gene regulatory networks has established a framework for a quantitative description of the dynamics of many important cellular processes. Such a description will require novel experimental techniques that enable the generation of time series data for the governing regulatory proteins in a large number of individual living cells. An ideal data acquisition system would allow for the growth of a large population of cells in a defined environment which can be monitored by high resolution microscopy for an extended period of time. Thus this lab will consist on a brief theory about microfluidics then will follow the practical work going from the chip fabrication to one of its applications: the tracking or monitoring of particles (beads or E. coli) in this device and the subsequent analysis of the acquired data. Some imaging techniques will also be introduced. Finally, a few questions will be discussed in order to outline some important points.
- Huh, Microfluidics for flow cytometric analysis of cells and particles, 2005
- Danino, A synchronized quorum of genetic clocks, 2010 (Chip reference)
- Youtube Commercial Lab on a Chip Device Presentation
Optical Trapping
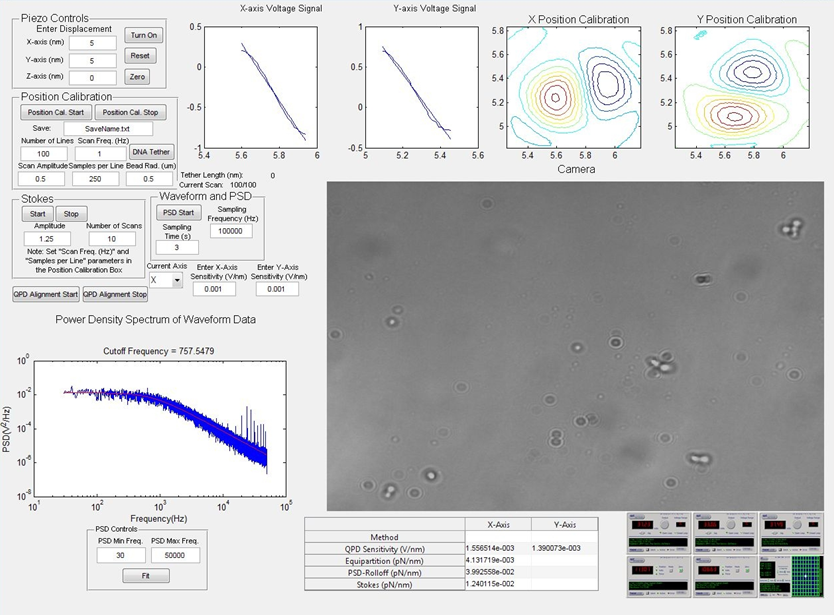 Optical trap is a powerfull tool used currently in many physical and biological applications. It allows for instance to manipulate and measurate with high precision different cellular tructure in biophysics and also to perform some experiments in biology such as cell sorting, single cell analysis…In this lab, our objectives are to become familiar with the fundamentals of optical trapping and to learn how to calibrate the optical traps for position detection and force measurement. Thus the sesson presents firstly a brief theory on optical trapping system and the controls for it. Then the subsequent practical work will be as following: Load a sample slide into the trap for calibrating the position detector, and find a bead attached to the coverglass surface. Run the Matlab position calibration to relate the voltage output of the position detector to bead displacement in nanometers. Make a new slide with free beads, find bead that is not attached to the surface and trap it. Use two different methods to calculate the trap stiffness and compare their results. These data will be analysed using Matlab.
Optical trap is a powerfull tool used currently in many physical and biological applications. It allows for instance to manipulate and measurate with high precision different cellular tructure in biophysics and also to perform some experiments in biology such as cell sorting, single cell analysis…In this lab, our objectives are to become familiar with the fundamentals of optical trapping and to learn how to calibrate the optical traps for position detection and force measurement. Thus the sesson presents firstly a brief theory on optical trapping system and the controls for it. Then the subsequent practical work will be as following: Load a sample slide into the trap for calibrating the position detector, and find a bead attached to the coverglass surface. Run the Matlab position calibration to relate the voltage output of the position detector to bead displacement in nanometers. Make a new slide with free beads, find bead that is not attached to the surface and trap it. Use two different methods to calculate the trap stiffness and compare their results. These data will be analysed using Matlab.
- Optical trapping lecture
- OT java applet 1
- Thorlabs optical tweezers
- MIT handout
- Ashkin, Forces of a Single-Beam Gradient Laser Trap on a Dielectric Sphere in the Ray Optics Regime, 1992
- Conroy, Optical waveguiding in suspensions of dielectric particles, 2005
- Youtube Optical Trapping Application on Beads
- Youtube Optical Trapping Application on Living Cells
Surface Design
Recent progress in microarray technology has importantly supported many fields such as medical diagonsis, food control, drug discovery, environment monitoring or toxicology research. This prominent success is partially due to the progress in the biomolecule detection and spotting technology. In this session, a “simplified DNA array” will be made. The exercise will explore a surface modification technique commonly employed in biosensor and microbiosensors. It will consist mainly of functionalizing a substrate surface by spotting manually oligonucelotide probes to detect DNA hybridization. So the session will review the very bases of surface preparation, functionalization as well as the data acquisition and analysis of such a biosensor. Understanding the control experiments is an essential objective of this practice as well.
Surface Plasmon Resonance
 Biosensors are widely used in different applications nowadays and different properties of materials and interfaces are used to design them. Among the biosensors currently used, some are based on optical properties in particular on the refractive index and thus the surface Plasmon resonance (SPR) phenomenon of the device interface. The surface Plasmon resonance based systems are very efficient because they enable the detection and quantification of biological interactions in real time, without the use of labels. They can be used to analyze samples going from low-molecular-mass drugs to multiprotein complexes and bacteriophages and finally, can also be used to detect interactions with very low affinity (from millimolar to picomolar in strength). In this laboratory, some aspects of Plasmon resonance based systems will be presented. Then the practical session will lead to detect β2μ-globulin using a SPR based system via two different strategies of ligands (probes) immobilization on the sensor chip: by amine coupling (direct immobilization) and using ligand capture method (indirect immobilization).
Biosensors are widely used in different applications nowadays and different properties of materials and interfaces are used to design them. Among the biosensors currently used, some are based on optical properties in particular on the refractive index and thus the surface Plasmon resonance (SPR) phenomenon of the device interface. The surface Plasmon resonance based systems are very efficient because they enable the detection and quantification of biological interactions in real time, without the use of labels. They can be used to analyze samples going from low-molecular-mass drugs to multiprotein complexes and bacteriophages and finally, can also be used to detect interactions with very low affinity (from millimolar to picomolar in strength). In this laboratory, some aspects of Plasmon resonance based systems will be presented. Then the practical session will lead to detect β2μ-globulin using a SPR based system via two different strategies of ligands (probes) immobilization on the sensor chip: by amine coupling (direct immobilization) and using ligand capture method (indirect immobilization).
Course Rules
1. Content
The teaching proceeds with weekly alternating sessions of laboratory practice and classroom data analysis. Both the laboratory practice and classroom data analysis sessions are supervised work in groups (3 people / group).
The core of this course consists of 6 exercises that are meant to be performed in either 2 weeks/sessions each for a total of 12 weeks:
- LAB-ON-A-CHIP: learn how to design and fabricate miniature chemical and bio-chemical analysis systems, also known as Labon-a-Chip systems, referring to the idea of shrinking a complete chemical analysis laboratory onto a small chip.
- BROWNIAN MOTION: learn how to how to simulate and analyze Brownian motion of single particles in Matlab, use brightfield and darkfield microscopy. They will be introduced to the image data acquisition, theory and software design for image filtering and particle tracking in Matlab.
- OPTICAL TRAPPING: learn the basics of operating a high-end optical tweezers to record mechanical transitions of single molecules.
- SURFACE DESIGN: learn some basic techniques of surface design for bioanalytics.
- SURFACE PLASMON RESONANCE: learn how to plan and interpret surface bio-molecular binding experiments
- ATOMIC FORCE MICROSCOPY: In this lab, you will learn the basics of atomic force microscopy, image single molecules as well as study the quaternary structure of collagen.
2. Evaluation
- 2/3: Written exam during exam session. The exam will contain the same kind of questions presented you in handouts.
- 1/6: Evaluation of the quality of the lab notebook. Student must compile properly the lab notebook.
- 1/6 Evaluation of the student’s preparation (before sessions) and participation (during sessions). Student has to study the handouts (provided on the web site) and prepare exercises beforehand. Before each exercise short quiz by TA will serve to asses the student preparation . If failed student doesn’t get any credits for this exercise however he/she is still obliged to do it !
3. Preparation
Students are asked to prepare lab sessions before the lab sessions, meaning understanding the theory and protocols, so that practical work can progress smoothly.
Students have to answer to questions from quiz (those one can answer without doing the experiment) so that they can discuss about with TA during sessions. Keep in mind that you will have the same or same kind of questions during the written exam; hence it is very important to really understand questions.
Lab Notebook
Keeping a complete and accurate record of experimental methods and data is a vital part of science and engineering. Your laboratory notebook is a permanent record of what you did and what you observed in the laboratory. Learning to keep a good notebook now will establish good habits that will serve you throughout your career.
A lab notebook should contain:
- Record of important procedures for experiments you have developed during your experimentation: everything you do, and why you did it, just like a diary!
- Record of your mistakes and difficulties. You will frequently learn more from these failures!
- Record of the results of experiments that you have performed.
- Enough information so that someone else, with an equivalent technical background to your own, is able to repeat your work, and obtain the same results by using your notebook
Lab notebook is not a:
- Copy of the handouts.
- Text book explaining the theory of phenomena you study or manual of machines.
- Simple list of results of experiments. Comment and draw conclusions!
What goes into your notebook? :
- Always use pen and write neatly and clearly
- Page numbers: in the upper outside corner of each page. These are important so you can refer back to frequently used tables, procedures, or results. You can also be sure that there are no missing pages (leading to missing steps) if following a past procedure.
- Table of contents: The first few pages should be reserved for this, it allows you to quickly find the information you are looking for and makes the book a useful reference. Later on you will be able to find a particular experiment without having to read every page.
- Dates: Every entry, or at the very least every day that you record data should be dated.
- When you start a new topic, start on a right side page (odd), and record its Title and Objectives at the top of the first page dedicated to this topic.
- Procedure of the experiment, all steps you have performed. Record Reasons for decisions made during an experiment as well. What we did isn’t always good enough, why we did what we did is just as important to record.
- Unusual conditions during an experiment. Something is went wrong or was unexpected.
- If you make a Mistake, don’t obliterate it! You may need to read your mistake later; perhaps you were right the first time! Use a single cross out and Explain why it was an error.
- Data typed into the computer must be printed and taped into your lab notebook. Plots of data made in lab should also be printed and taped in your lab notebook.
Safety
In any laboratory, there is potential for injury if certain common-sense practices are not followed. In AMBL this is minimal, but it’s still important to follow a few basic rules. In case of any doubt or question don’t hesitate to ask, and if you do some mistake REPORT IT IMMEDIATELY: doing mistakes is not an issue, you are here to learn, not reporting them is a big issue, since it can result in unexpected-unexplainable results, and even worst, can be harmful to you or for someone else in the lab. Always ask on how to dispose used stuff.
1. Electrical safety
Electrical injuries happen when large amounts of electrical power are dissipated by the body. Most often, this happens in high-current situations (mush more dangerous than the voltage!) and it’s a good idea to avoid becoming a current path.
In AMBL, we will work with only low-power electronics, and nothing we do is likely to cause injury. However, some common-sense precautions are in order:
- DO NOT connect supply voltages directly to ground
- DO NOT touch any current-carrying conductor with your bare hands
These simple rules will keep you from injuring yourself and damaging circuit components. Some components will have maximum power ratings that should not be exceeded, so pay attention to these values.
2. Laser safety
You will use a 300mW NIR diode lasers with λ=975nm during the Optical tweezers’ session. The hazards come from its higher power level, and because it is invisible, making it harder to be aware of its location/direction. The beam will be largely constrained in the apparatus, and you will not need to make adjustments that might put you in the beam path.
Please keep in mind the following things:
- Always know the path of the beam, and keep any body parts or reflective items (rings, watches, etc.) out of the beam path.
- Safety goggles will be available, but not required.
- Always read the pre-labs and know what special precautions you need to take associated with lasers or optics.
- When in doubt about doing something, don’t do it before checking with the lab instructor.
3. Chemical Safety & Biosafety
- DO NOT bring food or drink into the lab. The electronics will appreciate it.
- Always wear lab coat, gloves and goggles while working at the bench or at the chemical hood
- Dispose chemical/biological waste and sharps in proper containers.
- Wash your hands with soap and water after removing gloves and before leaving the lab.
- Please report any spills or injuries to the lab instructor immediately
Supplementary Materials
These following documents may also be useful for these practicals:
Teaching Assistant
…
Contacts
Professors:
Scientists:
TAs
- Arielle Louise Planchette(for SD)
- (for SD)
- (for SPR)
- (for SPR)
- Vytautas Navikas (for BM)
- Mukeshchand Thakur (for BM)
- Zoe Newell Swank (for LOC)
- Fabien Jammes (for LOC)
- Sebastian James Davis (OT)
- Evgenii Glushkov (OT)
- Samuel Mendes Leitão (AFM)