Dominique Bonvin, Michael Amrhein , Nirav Bhatt
Overview
Gas-liquid reaction systems such as hydroformylation, alkylation, carboxylation, polymerisation, hydrometallurgy and fermentation are key in producing industrial chemical, pharmaceutical and biochemical products. Models of such reaction systems are useful at the stages of process development in the laboratory and process operation in production. However, gas-liquid reaction systems are difficult to model because of the direct coupling between the chemical reactions and the transfer of reactants and products between the two phases. The proposed research deals with the development of a modeling framework for gas-liquid reaction systems and involves an incremental modeling approach using multi-sensor data. The first level consists in identifying features such as the time instants of reaction start/stop and whether a given reaction is mass-transfer controlled or not. This leads to a descriptive model. The second level involves the identification of a kinetic model consisting of a model structure and model parameters. Both levels are helpful in getting a better understanding of gas-liquid reaction systems. It is proposed to use information-rich spectroscopic and calorimetric data to build both the descriptive and the kinetic models in the laboratory during process development.
Keywords : Data analysis; modeling; gas-liquid reaction systems; homogeneous reaction systems; inlets; outlets; spectroscopic data; calorimetric data; multi-sensor data;

Laboratory Rector
Building a kinetic model involves the identification of a model structure (reaction stoichiometries, reaction-rate expressions) and of corresponding reaction-rate, thermodynamic and physical parameters from on-line and off-line experimental data obtained from multi-sensor data (such as spectroscopic data, calorimetric data, gas consumption data). This research aims at developing a modeling methodology for gas-liquid reaction systems that includes an incremental identification approach to obtain descriptive and kinetic models from the combined use (data fusion) of multi-sensor data. Figure 1 illustrates the various elements of the proposed modeling approach.
Concept of variants/invariants
The concentrations and the temperature (labeled original states) of reaction systems include information, which represents the combined effect of reactions (stoichiometry and kinetics), transport phenomena (mass-transfer rates, MTR), and the operation mode of the reactor (batch, semi-batch, continuous). A transformation is proposed, which expresses the original states as a linear combination of the initial conditions (known as reaction, MTR, and flow invariants) and three subsets of transformed states evolving with (i) reactions (known as reaction variants), (ii) mass-transfer rates (MTR) (known as reaction invariant and MTR variants), and (iii) flowrates (know as reaction and MTR invariants and flow variants).
This transformation has important implications for the analysis of gas-liquid reaction systems and for processing the corresponding pressure, calorimetric, concentration, and spectroscopic data.
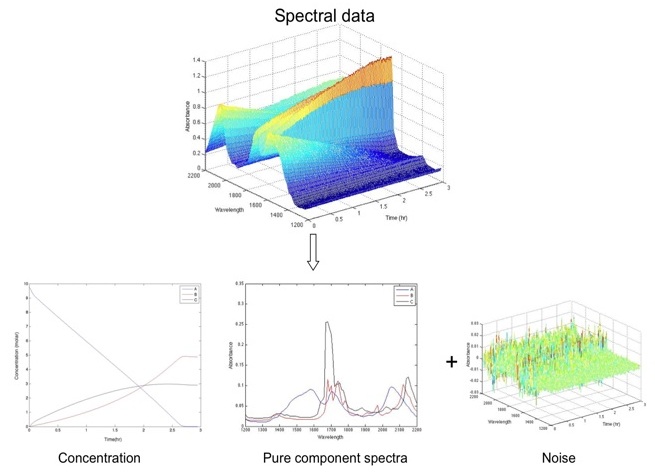
It is often assumed that a measured spectrum is a linear combination of the pure-component spectra of the absorbing species. In such cases, factor-analytical (FA) methods like multivariate curve resolution can be used for the calibration-free estimation of the concentration profiles based on spectroscopic, calorimetric and gas consumption data (see Figure 2). However, FA methods rely on appropriately formulated constraints and spectral matrices exhibiting an appropriate number of principal components (pseudo-rank).
For gas-liquid reaction systems, this research formulates such constraints from (i) ad-ditional information such as known/measured initial and final conditions, reaction pathway and reaction types (reversible, irreversible), and (ii) local rank information expressing process changes such start/stop of feeds and/or reactions, reaching of quasi-stationary conditions. FA methods can also take into account kinetic models as constraints and estimate kinetic parameters directly from spectroscopic, calorimetric and gas consumption data.
The pseudo-rank of the spectral matrix usually corresponds to the number of absorbing species. However, linear dependencies between concentration profiles resulting for example from underlying chemical reactions can lead to rank drop. FA methods fail if the pseudo-rank is less then the number of absorbing species (rank deficiency), and rank augmentation is required to break this rank deficiency.
Incremental identification
The objective of incremental identification is to identify the kinetic model and parameters for gas-liquid reaction systems. The incremental approach decomposes the identification task into a sequence of subproblems including (1) the estimation of selected concentration profiles using multivariate calibration (MVC) or multivariate curve resolution (MCR), (2) the identification of a stoichiometric model, (3) the estimation of reaction and mass-transfer rates, and (4) the identification of reaction and mass-transfer parameters through fitting the corresponding rate expressions.
Concentrations and heat effects are typically measured with some inaccuracy. Furthermore, the concentrations estimated from MVC and MCR also contain uncertainty that stems from measurement errors in the spectroscopic data and model errors in the MVC and MCR procedures. The incremental identification approach simplifies the estimation procedure since, at a given step, it does not require information regarding subsequent steps; however, it has the disadvantage of propagating model errors from step to step. This research studies the effect of error propagation in incremental identification.
Related publications
[epfl_infoscience url=”https://infoscience.epfl.ch/publication-exports/2862/”]